Research Article - Neuropsychiatry (2017) Volume 7, Issue 2
Mental health, pro-inflammatory cytokines, and brain derived neurotrophic factor in patients undergoing treatment for hepatitis C: comparison between peginterferon-alpha-2a and peginterferon-alpha-2b
- Corresponding Author:
- Chih-Ken Chen, M.D., Ph.D.
Department of Psychiatry, Chang Gung Memorial Hospital, Keelung, No.200 Lane 208, Ji-Jin 1st Road, Anle District, Keelung City 204, Taiwan
Tel: 886-2-24329292-2725
Fax: 886-2-24315931
E-mail: kenchen@ cgmh.org.tw
Abstract
Aim:
This study examines potential differences in the mental health, pro-inflammatory cytokines and brain-derived neurotrophic factor (BDNF) of patients with hepatitis C virus (HCV) infection who underwent antiviral treatment with peginterferon-alpha-2a (PegIFN-α-2a) and peginterferon-alpha-2b (PegIFN-α-2b).
Methods:
In a 24 week non-randomized longitudinal study, 22 patients with HCV were treated with PegIFN-α-2a plus ribavirin (Group A), and 12 patients were treated with PegIFN-α-2b plus ribavirin (Group B). All patients were assessed on the Hospital Anxiety and Depression Scale (HADS), the Short-form Health-related Quality of Life (SF-36) questionnaire, and their serum levels of interleukin-6 (IL-6), tumor necrosis factor-alpha (TNF-α), interleukin-1-beta (IL-1β) and BDNF were compared at baseline and weeks 4, 12 and 24.
Results:
During the 24-week antiviral treatment, Group A and Group B exhibited similar rates of sustained virological response, increased depression levels, decreased scores of physical component summary (PCS) of the SF-36 and reduced serum BDNF levels. However, increased serum levels of IL-6, TNF-α, and IL-1β were observed only in Group A at the 24th week. During the treatment period, the depression scores and PCS scores of the SF-36 were positively correlated with IL-6, TNF-α and IL-1β levels, and negatively correlated with BDNF levels. Anxiety scores of the HADS were positively correlated with TNF-α and IL-1β levels.
Conclusion:
This investigation provides insight into the differential effects of various peginterferon-alpha formulations on inflammatory parameters during antiviral treatment for HCV. During the treatment period, patients’ pro-inflammatory cytokines levels may be related to their symptoms of depression and anxiety, and BDNF levels may be linked to symptoms of depression. Future research with larger samples and a randomized control should be conducted to verify these findings.
Keywords
Hepatitis C, Cytokine, BDNF, Mood disorder, Quality of life, Interferon
Introduction
Hepatitis C viral (HCV) infection is a serious global public health concern worldwide. Globally, HCV affects around 130-210 million people [1], and the prevalence rate of anti- HCV in Taiwan has been estimated as 4.4% [2]. Patients with HCV infection are at risk of liver damage and hepatocellular carcinoma [3]. Interferon-alpha (IFN-α) has formed the basis of treatments for HCV infection, usually in conjunction with ribavirin (RBV) [4,5]. Two pegylated forms of IFN-α (PegIFN-α), PegIFN-α-2a and PegIFN-α-2b, differ in their pharmacokinetics and chemical properties, and both used with ribavirin, are recommended for the treatment of chronic HCV infection [6]. The goal of antiviral therapy is to eliminate the virus from the blood (sustained virological response, SVR), and to prevent complications of hepatitis C infection [7].
Numerous researchers have found that PegIFN- α-2a plus ribavirin yields a higher SVR and a lower discontinuation rate than PegIFN-α- 2b plus ribavirin [8-10]. However, a review article found that PegIFN-α-2a and PegIFN- α-2b exhibit similar degrees of effectiveness and tolerability, with few clinical differences [11]. Some recent investigations have also suggested similar SVR rates and frequencies of adverse events between PegIFN-α-2a and -2b [12,13]. To date, no conclusive evidence that one PegIFN-α is superior to the other has been obtained, and both forms of PegIFN-α plus ribavirin are considered to be standard treatments for chronic HCV infection.
Antiviral treatment with PegIFN-α and RBV can cause severe side-effects including fatigue, poor appetite, insomnia, and low blood count [14,15]. Moreover, depression is one of the most common neuropsychiatric complications during antiviral treatment against HCV infection [16,17]. A meta-analysis revealed that the overall cumulative incidence of depression is approximately 25-28% among patients with chronic HCV infection who are undergoing antiviral treatment for hepatitis C [18]. Depression is frequently associated with reduced quality of life (QOL), and poor adherence to, or early discontinuation of, treatment [19,20]. Therefore, understanding and detecting IFN-induced depression are crucial in clinical HCV treatment.
A complex pathophysiological background may be involved in IFN-induced depression [21]. Pro-inflammatory cytokines, particularly interleukin-6 (IL-6), tumor necrosis factor alpha (TNF-α) and interleukin-1 beta (IL-1β), have attracted increased attention in this respect [22,23]. Of these, IL-6 can act as both a proand an anti-inflammatory agent. These cytokines potentially change brain function (including neuroendocrine systems, neurotransmitters and apoptotic mechanisms) in several ways, and are associated with the pathogenesis of depression and impaired QOL [24]. However, causal relationships between mental health and pro-inflammatory cytokines require further clarification. Brain-derived neurotrophic factor (BDNF), an important growth factor in the central nervous system, is crucialfor the development, plasticity, and survival of neurons [25]. BDNF may mediate the detrimental effects of pro-inflammatory cytokines and depression by compromising neuroplasticity [26,27]. Compelling evidence demonstrates that BDNF in peripheral blood can be used as a biomarker for depression [28-30].
During antiviral treatment, IFN-α activates pro-inflammatory cytokines and contributes to the development of depressive symptoms [31]. A clinical study found that serum levels of BDNF declined in HCV patients who were undergoing 24 weeks of IFN-α treatment. Accompanying increased levels of pro-inflammatory cytokines were correlated with the development of depressive symptoms [32]. Some investigations have also suggested that IFN-induced depression is accompanied by immune activation and the reduction of serum BDNF levels [33,34]. The literature has demonstrated the similarity between the adverse effect profiles (including depression) of HCV patients who are treated with PegIFN- α-2a and -2b, although the rates of depressive symptoms vary among studies (6-65%) [35-37]. However, no investigation of whether PegIFN- α-2a and PegIFN-α-2b differently alter pro-inflammatory cytokine and BDNF levels has yet been published, and the relationships among patients’ QOL, pro-inflammatory cytokine and BDNF levels are poorly understood.
This study investigates the mental health status (depression, anxiety and QOL) and biochemical markers (pro-inflammatory cytokines and BDNF) of HCV patients who are undergoing a six-month antiviral treatment, and examines the potential differences between patients who were administered PegIFN-α-2a and PegIFN-α-2b.
Methods
▪ Participants and procedures
The Institutional Review Board of Chang Gung Memorial Hospital approved this study. Written informed consent was obtained from each patient before participation. Patients (aged ≥18 years) who were diagnosed with hepatitis C and were to receive antiviral treatment with PegIFN-α plus RBV at Chang Gung Memorial Hospital, Keelung, were enrolled. Patients were excluded from the study if they had a history of cancer, ischemic heart disease, co-infection with hepatitis B virus (HBV) or human immunodeficiency virus (HIV), psychotic disorders, organic mental disorders or substance use disorders. Thirty-four patients (with a mean age of 51.7±13.9 years, including 21 were females) met the inclusion criteria and received a 24-week follow-up with blood sample collection and mental health assessment.
This study was a 24-week, non-randomized, observational, prospective study. All 34 participants were administered subcutaneous PegIFN injections weekly and oral RBV daily for 24 weeks, consistent with the reimbursement criteria of the National Health Insurance in Taiwan. The treatment regimens were assigned at the discretion of the physicians. The prescribed PegIFN was PegIFN-α-2a, 180 μg, or weight-based PegIFN-α-2b, 1.5 μg/kg. For patients with genotype 1 HCV-infection, the oral RBV dose was 1000 mg per day for patients of body weight (BW) <75 kg, or 1200 mg for BW ≥75 kg. For patients with genotype non-1 HCV-infection, the RBV dose was 800 mg per day. All patients were followed up for 24 weeks until treatment had been completed. In the 24 treatment period, patients attended weekly outpatient visits for the first four weeks, biweekly visits between the 5th and 12th weeks, and monthly visits during the last 12 weeks. Haematological and liver biochemical tests were carried out in each visit. HCV RNA levels were measured before treatment (V1), at week 4 (V2), at week 12 (V3), and at week 24 (V4) of treatment. All biochemical and virological tests were conducted at the clinical laboratories of Chang Gung Memorial Hospital.
Of the 34 patients, 22 received antiviral treatment with PegIFN-α-2a plus RBV (Group A) while the remaining 12 were treated with PegIFN- α-2b plus RBV (Group B). The demographic characteristics and physical conditions of the two groups did not differ significantly. The second, third, and fourth study assessments were carried out at week 4, week 12, and week 24 after the beginning of the antiviral treatment. The procedure (blood sample collection and mental health assessment) that was conducted at baseline was repeated in each visit.
▪Mental health assessments
All patients underwent psychiatric diagnostic interviews of the Mini International Neuropsychiatric Interview (MINI) type, and their mental health statuses were assessed using the Hospital Anxiety and Depression Scale (HADS) and the Short-form Health-related Quality of Life (SF-36) questionnaire.
The MINI is a short structured diagnostic interview that is based on the statistical manual of mental disorders (DSM-IV) or International Statistical Classification of Diseases and Related Health Problems 10th Revision (ICD-10) [38]. This structured interview has high reliability and has been extensively used in international clinical studies. The inter-rater reliability of the Mandarin MINI, which was translated by the Taiwan Society of Psychiatry, is approximately 0.75 [39,40].
The HADS is a 14-item self-administered questionnaire for evaluating the severity of depression [41]. It is a commonly used assessment tool in hospital practice and primary care, and is also utilized for the general population. Seven of the items measure levels of anxiety and the other seven measure levels of depression. Each item is scored on a four-point Likert scale (0- 3), and the anxiety and depression subscales have independent measures. The validity of the Chinese version of the HADS has been established [42,43].
The SF-36 is an extensively used tool for assessing the health-related quality of life of patients [44]. The SF-36 incorporates eight subscales - physical functioning, physical role functioning, bodily pain, general health, vitality, social functioning, emotional role functioning, and mental health. The score ranges from 100 (highest) to 0 (lowest). A standard scoring algorithm aggregates scores into two summary scores, which are the physical component summary (PCS) and mental component summary (MCS) [45]. Lu et al. [46] validated the Chinese version of the SF-36, which has been widely used for measuring QOL in Taiwan [47].
▪ Laboratory tests
Venus blood from the participants was collected. Levels of aspartate transaminase (AST), alanine transaminase (ALT), alpha-fetoprotein, IL-6, TNF-α, IL-1β, BDNF, HCV RNA therein were measured in the laboratory. All of the blood samples were frozen after blood collection and stored at -80◦C until the laboratory measurements was made. AST, ALT and alpha-fetoprotein levels were determined using standard autoanalyzers. The serum levels of IL-6, TNF-α and IL-1β were obtained using the standard enzyme-linked immunosorbent assay (ELISA) protocol with a commercially available kit (eBioscience). The ranges of sensitivity of this method were 2-200 pg/mL for IL-6, 4-500 pg/mL for TNF-α, and 4-500 pg/mL for IL-1β, respectively. The serum BDNF levels were measured using an ELISA kit (Promega, Madison, WI, USA) following appropriate dilution of samples (1:10 to 1:50) in blocking and sample buffers, according to the manufacturer’s instructions [48]. BDNF levels were expressed as pg/ml, and the detection sensitivity of BDNF was 15.6 pg/ml.
Anti-HCV tests were conducted using a third-generation enzyme immunoassay kit (Abbott Laboratories, Berkshire, UK). A real-time polymerase chain reaction (PCR) assay (Roche Molecular Systems, Inc., Branchburg, NJ, USA), with a detection limit of 15 IU/mL, was used to quantify serum HCV RNA. HCV genotype was identified using a linear probe assay (Bayer Corporation, NY, USA). The HCV RNA load was examined before treatment, at four weeks and 12 weeks during treatment, at the end of treatment, and 24 weeks after treatment. The treatment efficacy was evaluated in a follow-up six months after combined pegIFN and RBV therapy. Patients who were HCV RNA-negative after six months during the follow-up were regarded as SVR.
▪ Statistical analysis
Data were analyzed using the Statistical Package for the Social Sciences for Windows (version 16.0; SPSS, Inc., Chicago, IL, USA). Variables are presented as either mean±standard deviation (SD) or frequency. A chi-squared (χ2) test or t test was performed to compare the characteristics of the two patient groups. All statistical tests were two-tailed, and differences were regarded as significant at p < 0.05.
A linear mixed model or generalized estimating equations (GEE) that were based the maximum likelihood estimation method and the auto-regression covariance matrix provided the primary means of analyzing the longitudinal data. These techniques handle missing data more effectively than do traditional methods (such as repeated-measure analysis of variance and ANOVA) [49,50]. The linear mixed model was used to elucidate the trends in mental health assessments, proinflammatory cytokines and BDNF level during the six months of follow-up, and LSD post hoc tests were conducted to compare multiple values. The GEE were used to elucidate the potential effects of treatment methods (Group B vs. Group A), proinflammatory cytokines level and BDNF level on the mental health status of patients during the 24 weeks of treatment period. Potential correlations between mental health status, cytokine level and BDNF level during the 24-week antiviral treatment were also estimated using GEE models. The depression, anxiety and quality of life scores were the dependent variables, and the treatment group, cytokine level and BDNF level were the independent variables.
Results
This study involved 22 patients who were undergoing antiviral treatment with PegIFN- α-2a plus RBV (Group A) and 12 patients who were receiving treatment with PegIFN-α- 2b plus RBV (Group B). Table 1 summarizes the demographic characteristics, biochemistry markers and mental health statuses of the two patient groups at the baseline. At the beginning of combination therapy, patients in Group B had a better physical QOL (PCS of the SF-36) than those in Group A (p = 0.017). The groups did not differ significantly with respect to the other variables.
| Characteristics | Group A (N=22) | Group B (N=12) | Statistical Value | P-Value |
|---|---|---|---|---|
| Sex (female/male) (n, %) | 14 (63.6) / 8 (36.4) | 7 (58.3) / 5 (41.7) | χ2= 0.092 | 0.761 |
| Age at recruitment (years) | 50.8 ± 15.6 | 53.3 ± 10.7 | t= 0.506 | 0.616 |
| Body weight (kg) | 68.5 ± 12.9 | 67.6 ± 14.9 | t= 0.191 | 0.849 |
| Education levels (years) | 7.7 ± 5.1 | 7.8 ± 4.3 | t = 0.056 | 0.956 |
| Employed (n, %) | 11 (50) | 7 (58.3) | χ2= 0.216 | 0.642 |
| Number of comorbid physical illnesses | 2.5 ± 1.8 | 2.3± 1.4 | t= 0.201 | 0.842 |
| HCV genotype (type 1/type 2 or 3) (n, %) | 11 (50) / 11 (50) | 6 (50) / 6 (50) | χ2 = 0.000 | 1.000 |
| Duration of being diagnosed hepatitis C (months) | 51.4 ± 56.7 | 27.7 ± 24.9 | t = 1.370 | 0.180 |
| AST/GOT levels (U/L) | 88.7 ± 20.7 | 79.6 ± 12.6 | t = 1.388 | 0.175 |
| ALT/GPT levels (U/L) | 134.6 ± 45.6 | 128.5 ± 44.1 | t = 0.376 | 0.709 |
| α-fetoprotein level (ng/mL) | 8.8 ± 7.9 | 10.0 ± 7.5 | t = 0.431 | 0.670 |
| Peginterferon-α-2a dose (mcg/week) | 173.9 ± 15.8 | - | N/A | N/A |
| Peginterferon-α-2b dose (mcg/week) | - | 98.2 ± 10.8 | N/A | N/A |
| Ribavirin dose (mg/day) | 1047.6 ± 125.0 | 983.3 ± 183.7 | t = 1.386 | 0.176 |
| Proinflammatory cytokines and BDNF | ||||
| Interleukin 6 (pg/mL) | 4.9 ± 7.4 | 6.6 ± 16.4 | t= 0.424 | 0.675 |
| Tumor necrosis factor α (pg/mL) | 2.0 ± 4.6 | 1.8 ± 3.7 | t = 0.131 | 0.897 |
| Interleukin 1β (pg/mL) | 4.7 ± 14.2 | 13.4 ± 40.2 | t= 0.917 | 0.366 |
| BDNF level (pg/mL) | 818.7 ± 120.2 | 858.2 ± 99.2 | t = 0.940 | 0.355 |
| Mental health status | ||||
| HADS-Depression score | 6.0 ± 4.2 | 4.1 ± 2.7 | t= 1.432 | 0.162 |
| HADS-Anxiety score | 6.8 ± 4.0 | 5.8 ± 4.5 | t = 0.631 | 0.533 |
| Physical component summary (PCS) of SF-36 | 60.6 ± 32.7 | 82.5 ± 17.8 | t = 2.530 | 0.017 |
| Mental component summary (MCS) of SF-36 | 62.1 ± 15.3 | 56.0 ± 19.5 | t = 1.012 | 0.319 |
Table 1: Baseline characteristics of patients with hepatitis C infection who received antiviral treatment using peginterferon-α-2a plus ribavirin (Group A) and those treated with peginterferon-α-2b plus ribavirin (Group B).
Of the 22 patients in Group A, 21 (95.5%), 19 (86.4%), and 13 (59.1%) remained in the study at weeks 4, 12, and 24, respectively. Of the 12 patients in Group B, ten (83.3%), seven (58.3%), and seven (58.3%) remained in the study at weeks 4, 12, and 24, respectively. After six months of follow-up, seven (58.3%) patients in Group A and five (57.1%) patients in Group B were SVR (HCV RNA-negative), respectively. The attrition rates (χ2 = 0.002, p = 0.966) and treatment response rates (χ2 = 0.003, p = 0.960) did not differ significantly between groups at the endpoint.
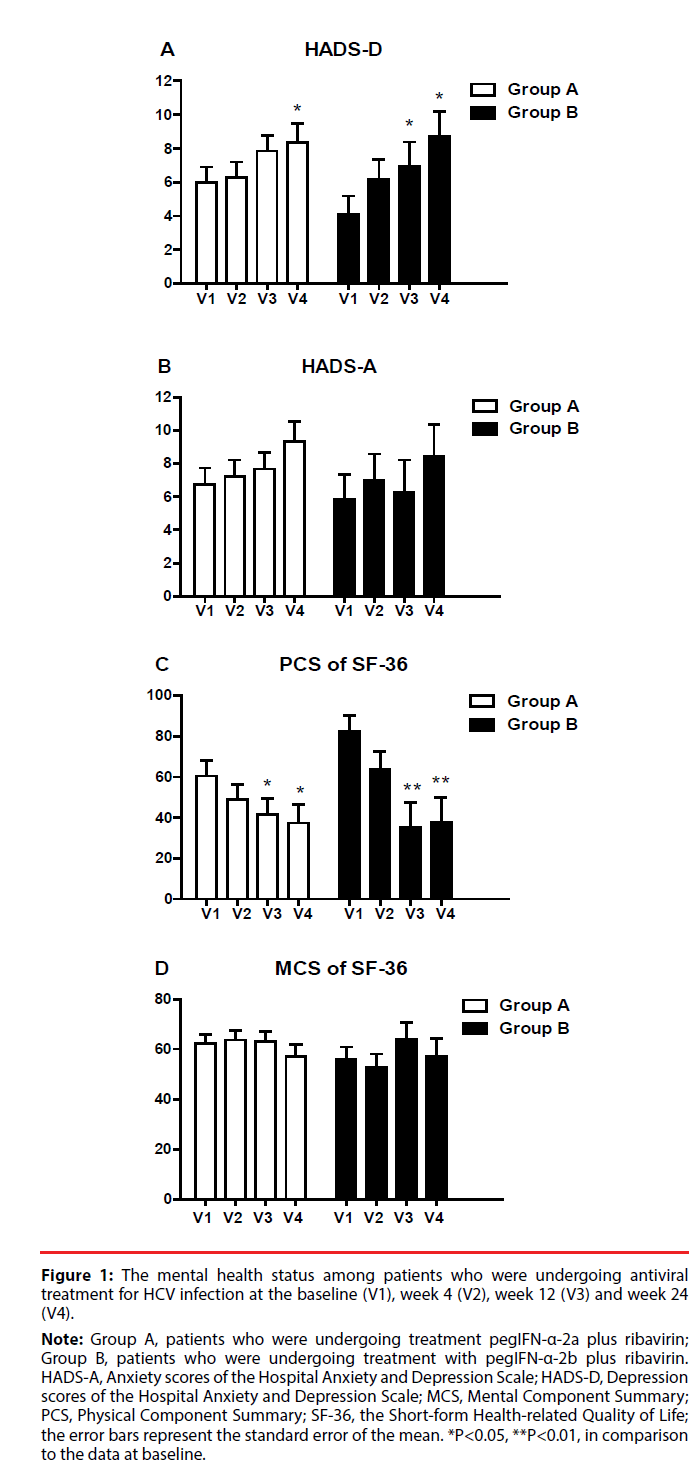
Figure 1 presents the changes in mental health status of patients with hepatitis C during the 24 weeks of treatment. For patients in Group A, the depression scores were significantly higher the 24th week of treatment than at the baseline (p = 0.045). For patients in Group B, depression scores were significantly higher at the 12th week (p = 0.045) and the 24th week (p = 0.028) than at the baseline. In both groups, the anxiety scores and the MCS scores did not significantly change over time.
Figure 1: The mental health status among patients who were undergoing antiviral
treatment for HCV infection at the baseline (V1), week 4 (V2), week 12 (V3) and week 24
(V4).
Note: Group A, patients who were undergoing treatment pegIFN-α-2a plus ribavirin;
Group B, patients who were undergoing treatment with pegIFN-α-2b plus ribavirin.
HADS-A, Anxiety scores of the Hospital Anxiety and Depression Scale; HADS-D, Depression
scores of the Hospital Anxiety and Depression Scale; MCS, Mental Component Summary;
PCS, Physical Component Summary; SF-36, the Short-form Health-related Quality of Life;
the error bars represent the standard error of the mean. *P<0.05, **P<0.01, in comparison
to the data at baseline.
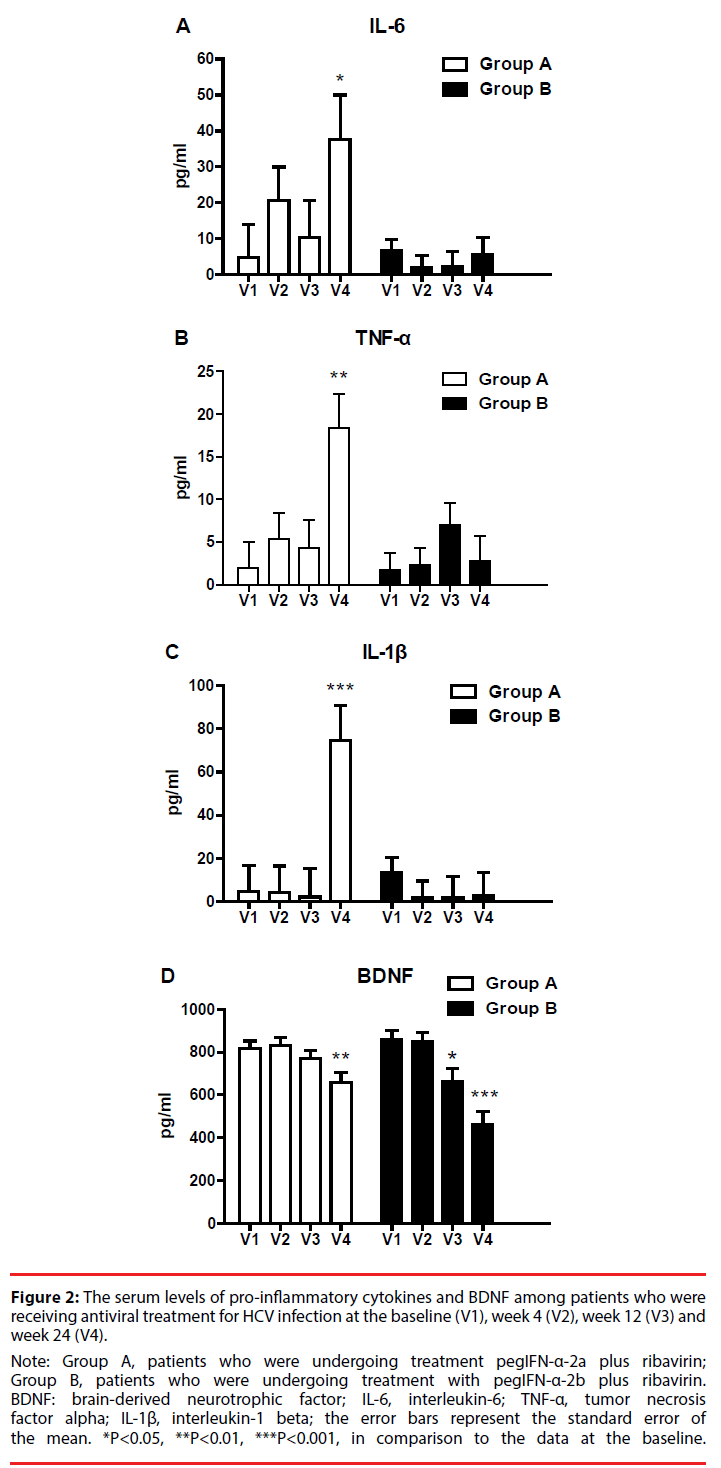
Figure 2 displays changes in pro-inflammatory cytokine and BDNF levels during the 24 weeks of treatment. For patients in Group A, the levels of IL-6 (p = 0.037), TNF-α (p = 0.002), and IL-1β (p = 0.001) were higher at the 24th week of treatment than at the baseline. For patients in Group B, the levels of IL-6, TNF-α, and IL-1β did not vary significantly over time. In Group A, plasma BDNF levels were significantly lower at the 24th week of treatment than at the baseline (p = 0.006). In Group B, BDNF levels significantly declined after the 12th week of treatment (week 12: p = 0.016; week 24: P < 0.001).
Figure 2: The serum levels of pro-inflammatory cytokines and BDNF among patients who were
receiving antiviral treatment for HCV infection at the baseline (V1), week 4 (V2), week 12 (V3) and
week 24 (V4).
Note: Group A, patients who were undergoing treatment pegIFN-α-2a plus ribavirin;
Group B, patients who were undergoing treatment with pegIFN-α-2b plus ribavirin.
BDNF: brain-derived neurotrophic factor; IL-6, interleukin-6; TNF-α, tumor necrosis
factor alpha; IL-1β, interleukin-1 beta; the error bars represent the standard error of
the mean. *P<0.05, **P<0.01, ***P<0.001, in comparison to the data at the baseline.
Table 2 shows the correlations between the mood symptoms, cytokine level and BDNF level of patients during 24 weeks of antiviral treatment, estimated using GEE models. The depression scores during treatment were positively related to IL-6 (B = 0.02, p = 0.016), TNF-α (B = 0.10, p = 0.035) and IL-1β (B = 0.03, p = 0.043) levels, and negatively correlated with plasma BDNF level (B = -0.01, p = 0.019). The anxiety scores during treatment were positively correlated with TNF-α (B = 0.14, p = 0.015) and IL-1β (B = 0.03, p = 0.036) levels. The treatment group (Group A or Group B) did not significantly affect the depression or anxiety of patients during the 24 weeks of treatment.
| Variables | Depression Scores of HADS | Anxiety Scores of HADS | ||||
|---|---|---|---|---|---|---|
| B (95%CI) | Wald χ2 | P-Value | B (95%CI) | Wald χ2 | P-Value | |
| Treatment (Group B vs. Group A) | -1.57 (-3.73, 0.59) | 2.042 | 0.153 | -1.21 (-3.78, 1.36) | 0.849 | 0.357 |
| Sex (female vs.male) | -0.09 (-2.23, 2.06) | 0.006 | 0.938 | 2.24 (-0.11, 4.59) | 3.478 | 0.062 |
| Age (years) | 0.02 (-0.06, 0.10) | 0.192 | 0.661 | -0.03 (-0.10, 0.05) | 0.498 | 0.480 |
| HCV genotype (type 1 vs. others) | 0.09 (-1.29, 1.47) | 0.016 | 0.899 | -0.09 (-1.50, 1.31) | 0.017 | 0.896 |
| Interleukin 6 (pg/mL) | 0.02 (0.004, 0.03) | 5.830 | 0.016 | 0.003 (-0.01, 0.01) | 0.297 | 0.586 |
| Tumor necrosis factor α (pg/mL) | 0.10 (0.01, 0.19) | 4.436 | 0.035 | 0.14 (0.03, 0.25) | 5.904 | 0.015 |
| Interleukin 1β (pg/mL) | 0.03 (0.001, 0.05) | 4.114 | 0.043 | 0.03 (0.002, 0.05) | 4.384 | 0.036 |
| BDNF level (pg/mL) | -0.01 (-0.01, -0.001) | 5.482 | 0.019 | -0.001 (-0.01, 0.01) | 0.109 | 0.741 |
Table 2: Relationships between patients’ mood symptoms, cytokines and BDNF levels of patients with hepatitis C during the 24-week antiviral treatment with peginterferon-alpha plus ribavirin.
Table 3 summarizes the correlations between patients’ health-related QOL, cytokine level and BDNF level during the study period. The PCS score was negatively correlated with patients’ age (B = -0.75, p = 0.008), and the levels of IL-6 (B = -0.17, p = 0.002), TNF-α (B = -1.13, P < 0.001) and IL-1β (B = -0.32, P < 0.001), and positively correlated with BDNF level (B = 0.05, p = 0.004). Patients in Group B had higher PCS scores than those in Group A (B = 18.98, p = 0.006) during the 24 weeks of treatment. Meanwhile, female patients had significantly lower MCS scores than did male patients (B = -12.85, p = 0.007). The antiviral treatment group, cytokine level and BDNF level were not significantly correlated with MCS score.
| Variables | Physical Component Summary of SF-36 | Mental Component Summary of SF-36 | ||||
|---|---|---|---|---|---|---|
| B (95%CI) | Wald χ2 | P-Value | B (95%CI) | Wald χ2 | P-Value | |
| Treatment (Group B vs. Group A) | 18.98 (5.48, 32.49) | 7.588 | 0.006 | -6.97 (-17.10, 3.15) | 1.821 | 0.177 |
| Sex (female vs.male) | 9.88 (-3.99, 23.74) | 1.950 | 0.163 | -12.85 (-22.26, -3.45) | 7.719 | 0.007 |
| Age (years) | -0.75 (-1.30, -0.20) | 7.105 | 0.008 | 0.12 (-0.21, 0.44) | 0.501 | 0.479 |
| HCV genotype (type 1 vs. others) | 6.33 (-4.50, 17.16) | 1.313 | 0.252 | -2.27 (-8.74, 4.21) | 0.470 | 0.493 |
| Interleukin 6 (pg/mL) | -0.17 (-0.28, -0.06) | 9.681 | 0.002 | 0.02 (-0.02, 0.06) | 0.966 | 0.326 |
| Tumor necrosis factor α (pg/mL) | -1.13 (-1.70, -0.55) | 14.690 | <0.001 | 0.07 (-0.18, 0.31) | 0.275 | 0.600 |
| Interleukin 1β (pg/mL) | -0.32 (-0.47, -0.18) | 18.261 | <0.001 | -0.05 (-0.11, 0.02) | 1.927 | 0.165 |
| BDNF level (pg/mL) | 0.05 (0.02, 0.09) | 8.423 | 0.004 | 0.00 (-0.01, 0.01) | 0.002 | 0.962 |
Table 3: Relationships between patients’ Health-Related Quality of Life, cytokines and BDNF levels of with hepatitis C during the 24-week antiviral treatment with peginterferon-alpha plus ribavirin.
Discussion
The results in this study indicate that 24 weeks of antiviral treatment with PegIFN-α-2a plus RBV or PegIFN-α-2b plus RBV yielded similar SVR rates, consistently increased depression level, reduced the physical component of QOL and reduced serum BDNF level. However, the serum levels of IL-6, TNF-α, and IL-1β only increased to the 24th week in patients who were administered pegIFN-α-2a, and not in those who were administered with PegIFN-α-2b. During the antiviral treatment period, depression scores and the physical component of QOL were positively correlated with IL-6, TNF-α and IL- 1β levels, and negatively correlated with plasma BDNF level.
▪ Antiviral treatment and mental health status
Compelling evidence has established that depression, anxiety and QOL become worse during antiviral treatment for HCV infection [16-18,31,51,52]. Twenty-four weeks of antiviral treatment increased the depression score and reduced the physical component of QOL, but did not significantly affect the anxiety score or mental component of QOL. Combination therapy with PegIFN-α plus RBV often has side-effects, such as fatigue, poor appetite, insomnia, and anemia [14,15]. These complications are associated with a decline in QOL, particularly in its physical component, mimicking the vegetative aspects of depression [19]. Therefore, the adverse effects of antiviral treatment may resemble depression or anxiety symptoms. Determining whether these mood symptoms are direct physiological effects or a psychological reaction to the combination therapy of PegIFN-α plus RBV is difficult.
Several articles have claimed that PegIFN-α- 2a provides a better outcome than PegIFN- α-2b [8-10], whereas many others report no difference in outcomes [11-13]. Although the relative efficacies of different forms of PegIFN-α are still debated, the results in current study reveal that neither SVR rate nor the variation of mental health status, differed between patients who were treated with PegIFN-α-2a (Group A) and those who were treated with PegIFN-α-2b (Group B). However, in this study, treatments were administered using non-randomized and open-label methods. Patients in Group B had a better physical QOL than those in the Group A at the baseline. Furthermore, the study population was female-dominant (61.8%). Although a previous large-scale investigation of a community in Taiwan revealed that the prevalence of HCV infection is slightly higher in females (6.2%) than in males (4.8%) [53], the gender distribution in the present study sample was not representative of the overall patient population. Therefore, the results might have been unwittingly affected by selection bias. Previous randomized trials have revealed that patients who are treated with PegIFN-α-2a plus RBV and those treated with PegIFN-α-2b plus RBV exhibit similar incidences of depression [35,36]. However, another study found that patients who were treated with PegIFN-α-2b plus RBV were more likely to develop depression than counterparts who were administered PegIFN- α-2a plus RBV [37]. Therefore, a study must be carried out to determine whether PegIFN-α variant differently alter mental health.
▪ Antiviral treatment, pro-inflammatory cytokines and BDNF
To the best of our knowledge, this investigation is the first to provide evidence of a relationship between the levels of pro-inflammatory cytokines and PegIFN-α species. Serum levels of IL-6, TNF-α, and IL-1β in Group A had increased by the 24th week of treatment, but those in Group B had not. This finding implies that various PegIFN-α formulations have different effects on immune response, and the administration of PegIFN-α-2a administration lad to a delayed activation of IL-6, TNF-α and IL-1β. Differences between these two PegIFN-α preparation include molecular weight, subtype of human IFN-α, pharmacokinetic profile (especially half-life) and pharmacodynamic gap [6,54,55]. PegIFN-α-2a is a long-acting interferon; is monopegylated with a 40kDa branched PEG moiety, and comprises four major positional isomers that involve Lys31, Lys121, Lys131, and Lys134 of interferon-α-2a [8-10]. We hypothesize that the unique pharmacological profile of PegIFN-α-2a may be related to the increase in serum levels of cytokines at week 24; however, the mechanism by PegIFN-α-2a and -2b differently alter pro-inflammatory cytokine levels warrants further study. Additionally, the small sample size and high attrition rate may have caused mean cytokine levels to be easily influenced by outliers. Therefore, the abrupt change in the cytokine levels in Group A at week 24 may also be associated with issues with the methodology of the study.
In the present study, reduced serum levels of BDNF were observed in both treatment groups. During the treatment for HCV, IFN-α can penetrate the brain parenchyma, and proliferate pro-inflammatory cytokines in certain areas of the brain [56]. Several investigations have demonstrated that inflammation can significantly reduce BDNF level within the brain by modulating BDNF gene expression [26,27]. In summary, we suspect that PegIFN-α-2a and PegIFN-α-2b similarly reduce BDNF levels during 24 weeks of treatment.
▪ Relationships among pro-inflammatory cytokines, BDNF and mental health
The potential causal relationship among response to antiviral treatment, changes in cytokine levels and depression may be complex and bidirectional. Consistent with the findings herein, some researchers have established that treatment with interferon-α has physical adverse effects with neuropsychiatric symptoms (such as depression) [16-18]. In contrast, some investigations have reported that degrees of depression and inflammation in HCV patients decline with as the virus is cleared (SVR) [57,58]. IFN is a member of the large class of cytokines, which can regulate the reproduction of cells and trigger the defenses of the immune system that help eradicate pathogens. We suggest that patients’ depression levels increased during the 24 weeks of combination therapy with PegIFN-α plus RBV, but variations in neuropsychiatric symptoms after the 24th week require further clarification.
In the treatment period, depression and the physical component of QOL were strongly correlated with serum levels of IL-6, TNF-α, IL- 1β, and BDNF. Anxiety scores during treatment were positively correlated with TNF-α and IL- 1β levels. These findings are consistent with those of an earlier clinical study [32], in which reduced serum levels of BDNF and increased cytokine levels were significantly correlated with the development of depressive symptoms during IFN-α treatment. These findings have two possible explanations. The first is that the increased cytokine levels and decreased BDNF levels are two aspects of the same phenomenon and that either cytokines or BDNF is involved in the pathogenesis of treatment-emergent depression. The second explanation is that cytokines and BDNF collaborate to cause depression in patients who are undergoing antiviral treatment (perhaps because BDNF acts as a mediator). Evidence shows that BDNF level may affect resiliency against the development of cytokine-related depression [34], and specifically against a subset of symptoms that are distinct from those that are affected by polymorphisms of the serotonin transporter gene (5-HTTLPR) and Val66Met BDNF [33]. Taken together, the findings in this study indicate that IFN-induced depression may be explained in part by changing immune activation and neuroprotective capacity, as reflected by decreases in serum BDNF following treatment with PegIFN-α. However, the complexity and causal relationship among mental health, pro-inflammatory cytokine levels and BDNF level require further clarification.
Limitations
This investigation has several limitations. First, the sample was small, especially in Group B, disfavoring the statistical power of the detection of potential differences in mental health between treatment groups, and limiting the ability of this study to identify changes in anxiety scores. Second, this study was a naturalistic observational study, rather than a randomized controlled trial. The sample sizes in the two treatment groups were unequal, and the results of this study may have been influenced by selection bias. Third, various important factors that may have potentially affected trends in the mental health and biochemical substrates of patients were not identified, including compliance with the PegIFN-α and RBV treatment regimen, the presence of infectious diseases, and the undergoing of concomitant drugs therapy (antidepressants). The analysis herein did not control for possible inflammatory processes other than HCV during the 24 weeks of observation. Therefore, whether these factors influenced the results of this study is uncertain. Fourth, previous investigations have found that polymorphisms of Val66Met BDNF [33] and 5-HTTLPR [59] are associated with depression during IFN-α treatment. Information about these genotypes of HCV patients is lacking, and so was unable to determine whether patients’ mental health and biochemical markers were affected by polymorphisms of Val66Met BDNF and 5-HTTLPR. Finally, recent advances in treatment against chronic HCV infection have been achieved through the application of directly acting antiviral agents [7], and these will reduce the interference between mental health status and pro-inflammatory cytokines in HCV patients who are undergoing antiviral therapy.
Conclusion
The findings in this study provide insight into the differential effects of various PegIFN-α formulation on mental health, pro-inflammatory cytokines and BDNF during antiviral treatment for HCV. Increased serum levels of IL-6, TNF-α, and IL-1β were observed in patients who were treated with PegIFN-α-2a plus RBV, but not in those who were treated with PegIFN-α-2b plus RBV. However, two regimens similarly worsened HCV patients’ depression and the physical component of their QOL, and reduced their serum BDNF levels. Future research with larger samples and a randomized control should be carried out to confirm the findings of this study.
Acknowledgments
This study was supported by the National Science Council, Taiwan (NSC 96-2314-B-182A-090- MY2). LJW and SWC are joint first authors and contributed equally to this manuscript.
Statement of Interest
The authors declare no conflicts of interest.
References
- Shire NJ, Sherman KE. Epidemiology of Hepatitis C Virus: A Battle on New Frontiers. Gastroenterol. Clin. North. Am 44(4), 699-716 (2015).
- Sievert W, Altraif I, Razavi HA, et al. A systematic review of hepatitis C virus epidemiology in Asia, Australia and Egypt. Liver.Int31(Suppl 2), 61-80 (2011).
- Lee MH, Yang HI, Yuan Y, et al.Epidemiology and natural history of hepatitis C virus infection. World. J.Gastroenterol 20(28), 9270-9280 (2014).
- McHutchison JG, Gordon SC, Schiff ER, et al.Interferon alfa-2b alone or in combination with ribavirin as initial treatment for chronic hepatitis C. Hepatitis Interventional Therapy Group. N. Engl. J. Med339(21), 1485-1492 (1998).
- Hauser G, Awad T, Brok J, et al.Peginterferon plus ribavirin versus interferon plus ribavirin for chronic hepatitis C. Cochrane. Database. Syst. Rev2(CD005441 (2014).
- Sullivan SD, Craxi A, Alberti A, et al.Cost effectiveness of peginterferon alpha-2a plus ribavirin versus interferon alpha-2b plus ribavirin as initial therapy for treatment-naive chronic hepatitis C. Pharmacoeconomics 22(4), 257-265 (2004).
- Nakamoto S, Kanda T, Shirasawa H, et al.Antiviral therapies for chronic hepatitis C virus infection with cirrhosis. World. J.Hepatol7(8), 1133-1141 (2015).
- Yang Z, Zhuang L, Yang L, et al.Efficacy and Tolerability of Peginterferon alpha -2a and Peginterferon alpha -2b, Both plus Ribavirin, for Chronic Hepatitis C: A Meta-Analysis of Randomized Controlled Trials. Gastroenterol. Res.Pract2013(1), 739029 (2013).
- Hauser G, Awad T, Thorlund K, et al.Peginterferon alpha-2a versus peginterferon alpha-2b for chronic hepatitis C. Cochrane. Database. Syst. Rev2(1), CD005642 (2014).
- El Sabaawy D, El-Haggar S, El-Bahrawy H, et al.A comparative study of variants of pegylated interferon alpha in treatment of chronic HCV patients. APMIS 123(6), 482-489 (2015).
- Foster GR. Pegylated interferons for the treatment of chronic hepatitis C: pharmacological and clinical differences between peginterferon-alpha-2a and peginterferon-alpha-2b. Drugs 70(2), 147-165 (2010).
- Sandoval-Ramirez JL, Mata-Marin JA, Huerta Garcia G, et al. Responses to peginterferon alfa-2a vs alfa-2b plus ribavirin in a Mexican population with chronic hepatitis C. J. Infect. Dev.Ctries9(3), 267-273 (2015).
- Pouresmaeeli M, Alavian SM, Keshvari M, et al.Efficacy and Tolerability of Peginterferon alpha-2a and Peginterferon alpha-2b in Iranian Patients With Chronic Hepatitis C. Hepat. Mon15(9), e30780 (2015).
- Horikawa N, Yamazaki T, Izumi N, et al.Incidence and clinical course of major depression in patients with chronic hepatitis type C undergoing interferon-alpha therapy: a prospective study. Gen. Hosp. Psychiatry25(1), 34-38 (2003).
- Mahajan S, Avasthi A, Grover S, et al.Role of baseline depressive symptoms in the development of depressive episode in patients receiving antiviral therapy for hepatitis C infection. J.Psychosom. Res77(2), 109-115 (2014).
- Schaefer M, Capuron L, Friebe A, et al.Hepatitis C infection, antiviral treatment and mental health: a European expert consensus statement. J.Hepatol57(6), 1379-1390 (2012).
- Lotrich FE, Rabinovitz M, Gironda P, et al.Depression following pegylated interferon-alpha: characteristics and vulnerability. J.Psychosom. Res63(2), 131-135 (2007).
- Udina M, Castellvi P, Moreno-Espana J, et al.Interferon-induced depression in chronic hepatitis C: a systematic review and meta-analysis. J. Clin. Psychiatry73(8), 1128-1138 (2012).
- Dan AA, Martin LM, Crone C, et al.Depression, anemia and health-related quality of life in chronic hepatitis C. J.Hepatol44(3), 491-498 (2006).
- Cozzolongo R, Porcelli P, Lanzilotta E, et al.The role of alexithymia in quality of life impairment in patients with chronic hepatitis C during antiviral treatment. Compr. Psychiatry60(1), 17-25 (2015).
- Baranyi A, Meinitzer A, Stepan A, et al.A biopsychosocial model of interferon-alpha-induced depression in patients with chronic hepatitis C infection. Psychother.Psychosom82(5), 332-340 (2013).
- Haapakoski R, Mathieu J, Ebmeier KP, et al.Cumulative meta-analysis of interleukins 6 and 1beta, tumour necrosis factor alpha and C-reactive protein in patients with major depressive disorder. Brain.Behav.Immun 49(1), 206-215 (2015).
- Stieglitz J, Trumble BC, Thompson ME, et al. Depression as sickness behavior? A test of the host defense hypothesis in a high pathogen population. Brain.Behav.Immun49(1), 130-139 (2015).
- Udina M, Navines R, Egmond E, et al.Glucocorticoid receptors, brain-derived neurotrophic factor, serotonin and dopamine neurotransmission are associated with interferon-induced depression. Int. J. Neuropsychopharmacol19(4)(2015).
- Autry AE, Monteggia LM. Brain-derived neurotrophic factor and neuropsychiatric disorders. Pharmacolog.Rev 64(2), 238-258 (2012).
- Numakawa T, Richards M, Nakajima S, et al. The role of brain-derived neurotrophic factor in comorbid depression: possible linkage with steroid hormones, cytokines, and nutrition. Front. Psychiatry5(1), 136 (2014).
- Calabrese F, Rossetti AC, Racagni G, et al.Brain-derived neurotrophic factor: a bridge between inflammation and neuroplasticity. Front. Cell.Neurosci8(1), 430 (2014).
- Molendijk ML, Spinhoven P, Polak M, et al.Serum BDNF concentrations as peripheral manifestations of depression: evidence from a systematic review and meta-analyses on 179 associations (N=9484). Mol. Psychiatry19(7), 791-800 (2014).
- Lopresti AL, Maker GL, Hood SD, et al.A review of peripheral biomarkers in major depression: the potential of inflammatory and oxidative stress biomarkers. Prog.Neuropsychopharmacol. Biol. Psychiatry48(1), 102-111 (2014).
- Bocchio-Chiavetto L, Bagnardi V, Zanardini R, et al.Serum and plasma BDNF levels in major depression: a replication study and meta-analyses. World. J. Biol. Psychiatry11(6), 763-773 (2010).
- Asnis GM, De La Garza R, 2nd. Interferon-induced depression in chronic hepatitis C: a review of its prevalence, risk factors, biology, and treatment approaches. J. Clin.Gastroenterol40(4), 322-335 (2006).
- Kenis G, Prickaerts J, van Os J, et al.Depressive symptoms following interferon-alpha therapy: mediated by immune-induced reductions in brain-derived neurotrophic factor? Int. J.Neuropsychopharmacol14(2), 247-253 (2011).
- Lotrich FE, Albusaysi S, Ferrell RE. Brain-derived neurotrophic factor serum levels and genotype: association with depression during interferon-alpha treatment. Neuropsychopharmacology 38(6), 985-995 (2013).
- Cicek IE, Cicek E, Kayhan F, et al.The roles of BDNF, S100B, and oxidative stress in interferon-induced depression and the effect of antidepressant treatment in patients with chronic viral hepatitis: a prospective study. J.Psychosom. Res76(3), 227-232 (2014).
- Manns MP, McHutchison JG, Gordon SC, et al.Peginterferon alfa-2b plus ribavirin compared with interferon alfa-2b plus ribavirin for initial treatment of chronic hepatitis C: a randomised trial. Lancet 358(9286), 958-965 (2001).
- Neri S, Pulvirenti D, Bertino G. Psychiatric symptoms induced by antiviral therapy in chronic hepatitis C: comparison between interferon-alpha-2a and interferon-alpha-2b. Clin. Drug.Investig26(11), 655-662 (2006).
- Fried MW, Shiffman ML, Reddy KR, et al.Peginterferon alfa-2a plus ribavirin for chronic hepatitis C virus infection. N. Engl. J. Med347(13), 975-982 (2002).
- Sheehan DV, Lecrubier Y, Sheehan KH, et al.The Mini-International Neuropsychiatric Interview (M.I.N.I.): the development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J. Clin. Psychiatry 59 (Suppl 20), 22-33 (1998).
- Kuo CJ, Tang HS, Tsay CJ, et al.Prevalence of psychiatric disorders among bereaved survivors of a disastrous earthquake in taiwan. Psychiatr.Serv54(2), 249-251 (2003).
- Chou FH, Chou P, Su TT, et al.Quality of life and related risk factors in a Taiwanese Village population 21 months after an earthquake. Aust. N Z J. Psychiatry38(5), 358-364 (2004).
- Olsson I, Mykletun A, Dahl AA. The Hospital Anxiety and Depression Rating Scale: a cross-sectional study of psychometrics and case finding abilities in general practice. BMC. Psychiatry5(1), 46 (2005).
- Hung CI, Liu CY, Wang SJ, et al.The cut-off points of the Depression and Somatic Symptoms Scale and the Hospital Anxiety and Depression Scale in detecting non-full remission and a current major depressive episode. Int. J. Psychiatry. Clin.Pract16(1), 33-40 (2012).
- Chen CK, Tsai YC, Hsu HJ, et al. Depression and suicide risk in hemodialysis patients with chronic renal failure. Psychosomatics 51(6), 528-528 (2010).
- Ware JE Jr,Sherbourne CD. The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection. Med. Care30(6), 473-483 (1992).
- Taft C, Karlsson J, Sullivan M. Do SF-36 summary component scores accurately summarize subscale scores? Qual. Life. Res10(5), 395-404 (2001).
- Lu JF TH, Tsai YJ. Assessment of Health-related Quality of Life in Taiwan (I): Development and Psychometric Testing of SF-36 Taiwan Version. Taiwan. J. Pub. Health(2003).
- Tseng HM, Lu JF, Gandek B. Cultural issues in using the SF-36 Health Survey in Asia: results from Taiwan. Health. Qual. Life. Outcomes1(1), 72 (2003).
- Kurita M, Nishino S, Kato M, et al.Plasma brain-derived neurotrophic factor levels predict the clinical outcome of depression treatment in a naturalistic study. PLoS. One7(6), e39212 (2012).
- Kenward MG, Lesaffre E, Molenberghs G. An application of maximum likelihood and generalized estimating equations to the analysis of ordinal data from a longitudinal study with cases missing at random. Biometrics 50(4), 945-953 (1994).
- Zhang H, Paik MC. Handling missing responses in generalized linear mixed model without specifying missing mechanism. J.Biopharm. Stat19(6), 1001-1017 (2009).
- NaymanAlpat S, Usluer G, Yavuz H, et al.Effect of pegylated interferon treatments for chronic active hepatitis C on quality of life. J. Chemotherapy20(1), 101-105 (2008).
- Mm B, A Y, U Y, et al.Major depressive disorder and generalized anxiety disorder and response to treatment in hepatitis C patients in Egypt. Int. J. Psychiatry. Med50(2), 147-162 (2015).
- Lee MH, Yang HI, Jen CL, et al.Community and personal risk factors for hepatitis C virus infection: a survey of 23,820 residents in Taiwan in 1991-2. Gut 60(5), 688-694 (2011).
- Grace MJ, Cutler DL, Bordens RW. Pegylated IFNs for chronic hepatitis C: an update. Expert.Opin. Drug.Deliv 2(2), 219-226 (2005).
- Bruno R, Sacchi P, Ciappina V, et al.Viral dynamics and pharmacokinetics of peginterferon alpha-2a and peginterferon alpha-2b in naive patients with chronic hepatitis c: a randomized, controlled study. Antivir.Ther 9(4), 491-497 (2004).
- Guan Z, Fang J. Peripheral immune activation by lipopolysaccharide decreases neurotrophins in the cortex and hippocampus in rats. Brain.Behav.Immun20(1), 64-71 (2006).
- Bonkovsky HL, Woolley JM. Reduction of health-related quality of life in chronic hepatitis C and improvement with interferon therapy. The Consensus Interferon Study Group. Hepatology 29(1), 264-270 (1999).
- Boscarino JA, Lu M, Moorman AC, et al.Predictors of poor mental and physical health status among patients with chronic hepatitis C infection: the Chronic Hepatitis Cohort Study (CHeCS). Hepatology 61(3), 802-811 (2015).
- Lotrich FE, Ferrell RE, Rabinovitz M, et al.Risk for depression during interferon-alpha treatment is affected by the serotonin transporter polymorphism. Biol. Psychiatry65(4), 344-348 (2009).