Research Article - Neuropsychiatry (2016) Volume 6, Issue 1
Gender specific differences in auditory brain stem response in young patients with ADHD
- Corresponding Author:
- Emma Claesdotter-Hybbinette
Department of Clinical Sciences, Lund, Child and Adolescent Psychiatry, Lund University, Sofiavägen 2D, SE-22241 Lund, Sweden
Tel: +0046 768871765
Abstract
Objective:The auditory brainstem response (ABR) is often affected in neurodevelopmental disorders. The aim of this study was to investigate gender differences in ABR between young females and young males with ADHD, compared to control subjects.
Method:We studied 63 females with ADHD (mean 13.8years), 26 female controls (mean 13.8years), 48 males with ADHD (mean 13.1 years), and 20 male controls (mean 12.8years). All patients were diagnosed according to the DSM-IV. An ABR consists of seven positive peaks (wave I–VII) 10 ms following a stimulus, recorded by electrodes on the mastoid processes of each ear and on the forehead.
Results: When analysing the ABRs of the female ADHD patients 3 traits were identified; TR6, TR14 and TR15. The higher value in TR6 (p=0.000064) is explained as an aberrant thalamus profile. In TR14 (p=0.00059) presence of 3500 Hz-frequencies in the region from superior olivary complex to thalamus. TR15 (p=0.00035) represents more aberrant curve profiles in the region of the lateral leminiscus. In the ABR of the male patients we found we 3 traits; TR4, TR5 and TR14. TR 4 (p=0.00105) is a lower correlation to a norm curve in inferior colliculus and thalamic area. TR5 (p=0.00027) identifies irregular curve profiles representing the nucleus cochlea. TR14 (p=0.00013) presence of 3500 Hz-frequencies in the region from superior olivary complex to thalamus.
Conclusion: Young females with ADHD exhibited a significantly different ABR in a region between cochlear nucleus and superior olivary complex and in the thalamic region. In the male ADHD group ABR aberrancies were found in the midbrain region and in the more peripheral part; nucleus cocleus. The only trait that was significantly different between the ADHD group and the control subjects, for both male and females, was TR14. These data indicate both gender specific aberrations in the ABR in ADHD subjects as well as specific differences between ADHD subjects and normal controls.
Keywords
ABR; ADHD; Child and adolescent psychiatry; Diagnostics; Gender; Young patients
Introduction
Attention deficit hyperactivity disorder (ADHD) has been recognized as one of the most common neurodevelopmental disorders in childhood [1]. It is a heterogeneous condition with persistent symptoms of hyperactivity, inattention and impulsiveness which impair functioning in multiple settings [2,3]. ADHD is a highly heritable disorder and twin studies have shown a heritability rate of approximately 80 % [4]. The estimated worldwide prevalence of ADHD in children is around 5% [5]. The symptoms frequently persist into adulthood and are associated with functional limitations as well as psychiatric and somatic morbidity. ADHD is a considerable burden to the affected individual and to society [1,6]. ADHD is affecting both sexes, with a male to female ratio approaching 1:1 [7] contrasting to previous studies showing a gender ratio (girl:boy) ranging from 1:3 to 1:16 [8]. Over the years research has been trying to find differences in symptoms to explain the gender differences in prevalence. It has been proposed that girls with ADHD may be more likely to have the inattentive type of ADHD and may suffer more from internalizing symptoms and inattention while, boys on the other hand have more hyperactive and aggressive symptoms [9-12].
An ADHD diagnosis should be based on neurodevelopmental and clinical history [1,2]. In addition, neuropsychological testing and rating scales are often used as a supplement. There is a great need for objective measures to improve the diagnostic accurancy and to guide clinical interventions. There are recent studies that have focused on trying to find objective neuropsychological testing methods for the diagnostics of ADHD [13-17].
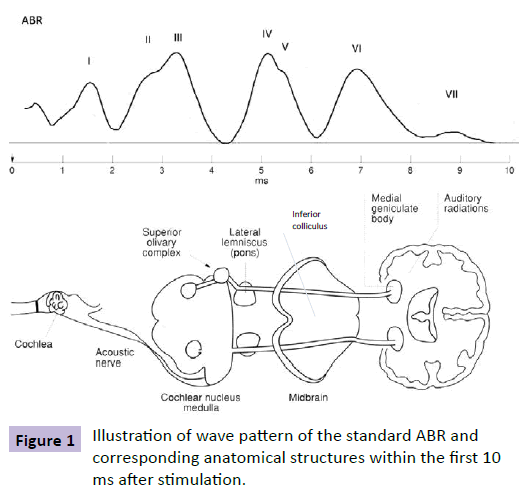
Auditory brainstem response (ABR) was first described in 1971 [18] and refers to the particular kind of event related potential (ERP) where the stimulus is sound, in this case, in the shape of distinct clicks at given intervals. In ABR, electrodes are placed and calibrated to study the processes that occur in the basic auditory pathways, situated in the brainstem [19] (Figure 1). ABR reflects the subcortical neuronal electrical activity in the auditory pathway within 10 milliseconds (ms) after brief auditory stimuli.
The ABR wave-pattern provides information in terms of the latencies and amplitudes of these peaks. Analysis of the ABR wave patterns normally comprises measurements of interpeak latencies as well as ratios of peak amplitudes [20,21]. ABR is an objective method that does not require active patient participation and is considered an objective approach to investigate brainstem function. In addition, complex stimuli may reveal aberrations, which may not be assessed by standard audiological ABR procedures. Complex click stimuli (e.g. forward masking) were therefore used in the present study to increase the possibility of detecting variances in comparison with matched healthy children. The importance of using complex stimuli is also stated for autism spectrum disorder, ASD [22].
Several studies have shown that ABR is often affected in ASDs [23-28] adults with ADHD [29,30], schizophrenia [28,31,32] and bipolar disorders [33]. Possible ABR abnormalities in young patients with ADHD need more studies [34,35].
We have previously presented a study on ABR in young females with ADHD where we found ABR traits specific for young females with ADHD compared to control subjects [14]. The aim of the present study is to investigate possible gender differences in ABR between young females and young males with ADHD, compared to control subjects.
Methods
Subjects
This study included a total of 63 females with ADHD (age mean 13.8 years, SD 2.5), 26 female control subjects (age mean 13.8 years, SD 2.7), 48 young males with ADHD (age mean 13.1 years, SD 1.8), and 20 male control subjects (age mean 12.8 years, SD 1.7). Patients and control subjects were in the age range of 7-17 years. The age difference between boys and girls were not statistically significant. All patients were instructed to not take any medication the day of the testing.
All patients were recruited from the Child and Adolescent Psychiatry outpatient department of Eslöv and Lund, two cities in the south of Sweden. All patients were diagnosed according to the Diagnostic and Statistical Manual of Mental Disorders, 4th Edition (DSM-IV). The diagnoses were confirmed by the same senior psychiatrist. The control groups were recruited from schools in Lund and recruited subjects had no previous record of any psychiatric disorder. Patients with other concurrent psychiatric diagnoses were excluded to avoid comorbidity. Subjects with hearing impairment were excluded from the study.
Written informed consent was obtained from all the subjects and their parents/guardians. The study was approved by the regional ethics committee at the Lund University (Dnr: 2010-120).
Apparatus and stimulus
The evoked potentials were recorded using auditory brainstem response, ABR. The ABR consists of a sequence of seven positive peaks (wave I–VII) that normally occur within 10 ms following the onset of a stimulus recorded by surface electrodes on the mastoid processes of each ear and on the forehead. Waves I and II are produced by the auditory nerve, whereas the subsequent peaks are due to the combined electrical activity of nuclei at gradually higher levels of the ascending auditory pathway in the brainstem. Waves III and IV are believed to be generated in the cochlear nucleus and superior olivary complex (SOC), respectively, whereas wave V is thought to represent activity at the levels of lateral lemniscuses and inferior colliculus [36,37]. The measuring system used was SensoDetect® BERA (Brainstem Evoked Response Audiometry) A1000. The stimuli were presented via TDH-50P headphones with Model 51 cushions (Telephonics, Farmingdale, New York, USA). Presentations were made binaurally with the stimuli in phase over headphones.
In total, 4 sound stimuli were used. The sound stimuli included square-shaped click pulses, high frequency varied pulses, forward masking and backward masking stimuli. The click pulses were repeated until a total of 1024 accepted evoked potentials had been collected for each sound stimulus. Thus, each ABR waveform represents an average of the responses to 1024 stimulus presentations. TTL (transistor-transistor logic) trigger pulses coordinated the sweeps with the auditory stimuli. A TTL pulse is the signal which tells the ABR system to measure. With a correctly timed TTL pulse, all ABR representations will be synchronized. Aberrant activity, such as extremely high amplitudes due to extraordinary movements was rejected. Sound levels were calibrated using a Bruel and Kjaer sound level meter and Type 4152 artificial ear (Bruel & Kjaer S & V Measurement, Naerum, Denmark). The acoustic output from the earphones corresponded to SPL: 80 dB HL or 109 peSPL (peak equivalence). A square-shaped click pulse was used as probe in the auditory masking stimuli [32]. The sound stimuli included square-shaped click pulses (0.136 ms duration, including 0.023 ms rise and fall; 192 ms interstimulus interval), high pass filtered pulses (a Butterworth high-pass filtered square shaped click pulse with a cutoff of 3000Hz), forward masking (12.3 ms gap from masker to click pulse) and backward masking (12.3 ms from click pulse to the masker) stimuli as previously described. A 1500-Hz Butterworth low-pass filtered white noise, with 15 ms duration (including 0.4 ms rise and fall times) was used as masker for both forward and backward masking stimuli. All stimuli were constructed using MATLAB Signal Processing Toolbox (The MathWorks, Inc., Natick, Massachusetts, USA) and stored in a flash memory in the SensoDetect®BERA system.
Procedure
All tests were performed in a soundproof slightly darkened room. Participants were comfortably seated in an armchair with their legs on a small footstool to assure a resting position. Five surface electrodes were applied: two reference electrodes on the mastoid bone behind the left and right ear, respectively, and two active electrodes and one ground electrode placed on the forehead. To make sure a good transmission the sites were washed with disinfectant and abrasive paste was used to stick the electrodes. Absolute impedances and inter electrode impedances were measured before and after the experiments to verify that electrode contact was maintained (below 5000 Ω). Earphones were fitted to cover both ears. The subjects were instructed to turn off their cellular phones and relax with their eyes closed and were permitted to fall asleep. The test requires no active participation other than being subjected to sound stimulation. Before the test situation written information had been sent home to the subject’s parents or guardians as well as to the subject. On site of the test session, subjects were again verbally informed of the nature of the experiments. The click sounds were presented to the subjects beforehand to make them acquainted with stimuli. The subjects were tested one at a time and the duration of the testing procedure was approximately 30 minutes.
Data analysis
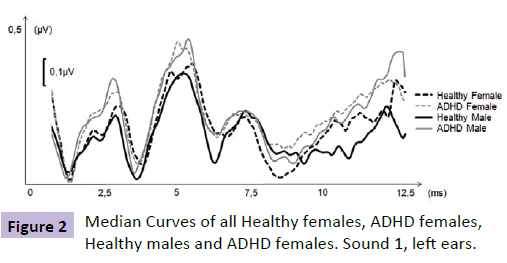
Prior to further analysis the audiogram was correlated to ABR data, from a group of healthy and normal hearing individuals, derived from a normative database to depict general audiogram quality. This is a standard operating procedure of this method in order to grant audiogram quality. A low correlation led to exclusion of the patient due to risk of erroneous measurement (e.g. loose electrodes or head phones). The Sensodetect system rejected all evoked potentials being of abnormally high voltage (i.e. aberrant activity), typically trigged by patient movement, coughing or tension. Collected evoked potentials for each sound stimulus from each individual was imported to Microsoft Excel (Microsoft Corp, Redmond, WA, USA) and analyzed using SensoDetect® BAS. Frequencies, latencies, amplitudes and correlation coefficients to a normed ABR curve, respectively, were investigated. Amplitudes were measured from the positive peak of a given wave to the bottom of the previous wave. Since the amplitude values obtained were not read in μV, microvolt outputs were indexed (i.e., normalized by adding constants to avoid negative values, then all amplitude values were divided by the highest observed amplitude). Thus, relative linear amplitudes are used in this study. In order to identify specific pathologies along the auditory pathway, correlation values to a normative ABR curve were calculated for different sections of the ABR. Values ranging from -1 to +1 were obtained using Spearman rho. High, positive values indicate similarity (e.g. no pathology) values around zero indicate no relation, and low values close to -1 indicate inverse relationship. After the r-values for all sections of the total ABR curve (0-10 ms) had been computed, the results were ranked. Thus, the test subject’s most aberrant ABR region, when compared with the norm curve, depicts a high number, and vice versa (Figure 2).
The same principle was used in order to identify occurrence of high frequencies in the ABR curves of the test subjects. A mathematically constructed artificial ABR (a sine wave with the frequency 3500 Hz) was used as norm. Norm population median values for every data point in the ABR were used to construct the artificial ABR. Every patient’s ABR curve was correlated to this artificial ABR. The correlation value was calculated for each possible starting point in the ABR and the r (max) was used to indicate occurrence of the specific frequency. As the ambition with this operation was to see whether the test person had an occurrence of the frequency or not, the outcomes were ranked from 1 for all values between r=-1 to r=+0.1 and thereafter 2 for r=0.1 to r=0.2 and so forth, with 10 for r=0.9 to r=1, indicating a perfect match to the normed ABR.
Aberrancies in the ABR were denoted traits (TR). The ABR trait numbering emanated from the fact that a higher number of potentially interesting traits were originally investigated. However, not all of these qualified in terms of test-retest prediction values. Thus, the traits presented in this study shows gaps in numbering.
For measuring differences between young males with ADHD compared to age matched control group and young females with ADHD compared to age matched control group, the nonparametric test Mann–Whitney U was used.
Results
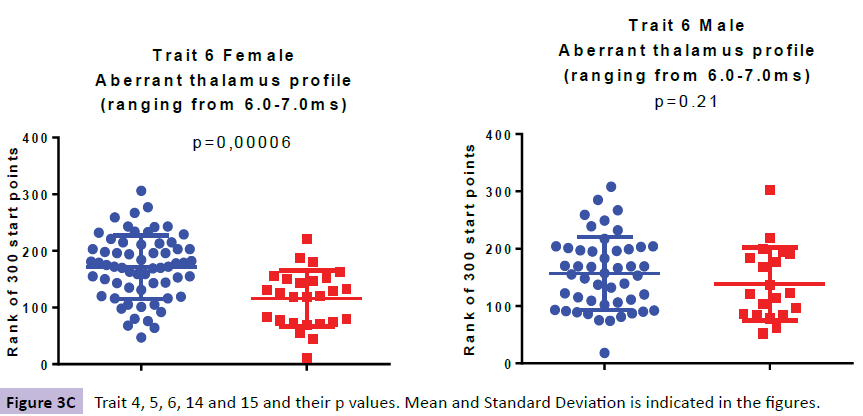
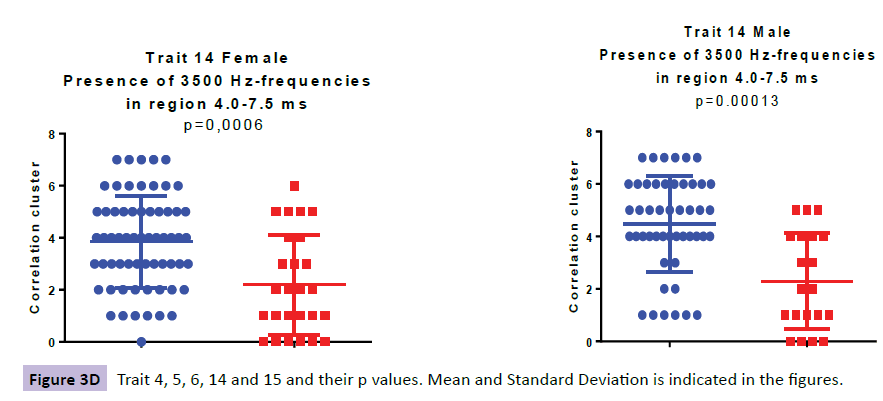
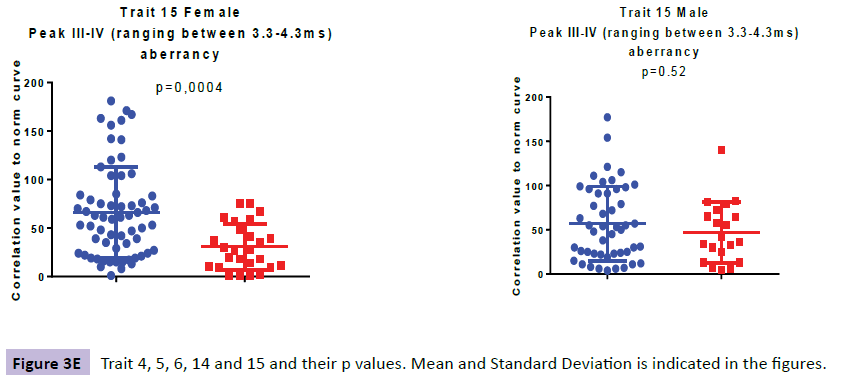
When analysing the ABRs of the female ADHD patients we found aberrancies in three wave areas these included wave VI (i.e. denoted TR6: Aberrant thalamus profile ranging from 6.0-7.0ms), wave IV-VI (i.e. denoted TR14: Presence of 3500 Hz-frequencies in the region 4.0-7.5ms ranging from SOC to thalamus) and waves III-IV (i.e. denoted TR15: Aberrant profile in the region 3.3-4.3ms ranging from cochlear nuclei to SOC). In the ABR of the male patients we found aberrancies in three areas; wave IV-VI (i.e denoted TR4: Low correlation to norm curve in the region 3.5-7.5 ms ranging from inferior colliculus to thalamus and i.e denoted TR14: Presence of 3500 Hz-frequencies in the region 4.0-7.5ms ranging from SOC to thalamus) and wave III (i.e denoted TR5: Low backward masking correlation to norm curve in the region 2.5-4 ms indicating a peripheral function deficit in the early auditory pathway.
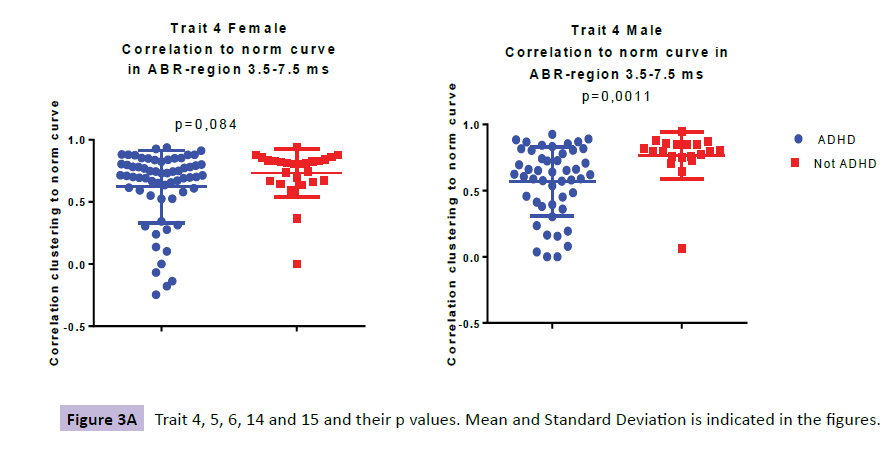
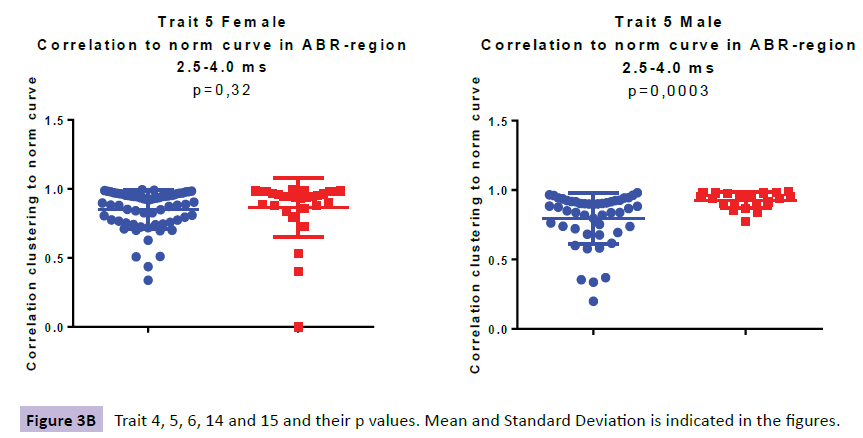
Comparing the ABR of 63 girls with ADHD to 26 age correlated control subjects three traits were identified, denoted TR6, TR14 and TR15 (Figure 3a-e).
The higher value in TR6, specific for the ADHD females (p=0.000064), as compared with the female controls, is explained by more aberrant curve profiles in the thalamic region at 6.0-7.0 ms. In TR14, a higher presence of 3500 Hz-frequencies in the midbrain region at 4.0-7.5 ms (ranging from SOC to thalamus), was observed for the ADHD group as compared with controls (p=0.00059). The higher value in TR15, specific for the ADHD females at 3.3-4.3 ms (p=0.00035), as compared with the female controls, is explained by more aberrant curve profiles in the region of the lateral leminiscus (Table 1).
| Trait | Female ADHD mean (S.D.) | Female controls mean (S.D.) | Female p-value | Male ADHD group mean (S.D.) | Male controls mean (S.D.) | Male p-value |
|---|---|---|---|---|---|---|
| median | median | |||||
| TR4 | 0.62 (0.29); 071 | 0.73 (0.19); 0,81 | 0.084 | 0.57(0.26); 0,62 | 0.77 (0,18); 0.80 | 0.00105 |
| TR5 | 0.85 (0.14); 0.90 | 0.85 (0.22); 0.94 | 0.321 | 0.80 (0.18); 0.87 | 0.93 (0,06); 0.95 | 0.00027 |
| TR6 | 172 (56); 174 | 116 (49); 123 | 0.000064 | 157 (63); 156 | 139 (64); 122 | 0.208 |
| TR14 | 3.8 (1.8); 4 | 2.2 (1.9); 2 | 0.00059 | 4.5 (1.8); 5 | 2.3 (1.8); 2 | 0.00013 |
| TR15 | 66 (47); 61 | 31 (24); 27 | 0.00035 | 57 (42); 52 | 47 (34); 39 | 0.518 |
Tabel 1: ABR results for young patients with ADHD compared to controls. Mann-Whitney U test was used. Female ADHD group (N=63), female controls (N=26), male ADHD group (N=48) and male controls (N=20).
When looking at the ABR from 48 young males with ADHD and comparing them to their 20 age correlated control subjects we found three traits; TR4, TR5 and TR14 (Figure 3a-e). TR4 is described as a lower correlation to a norm curve in inferior colliculus and thalamic area at 3.5-7.5 ms (p=0.00105). TR 5 identifies irregular curve profiles within peak III, ranging from at 2.5-4.0 ms, representing the cochlear nucleus (p=0.00027). TR14 is described as a higher presence of 3500 Hz-frequencies in the region at 4.0-7.5ms (ranging from SOC to thalamus), was observed for the ADHD male group as compared with controls (p=0.00013).
The only trait that was significantly different between the ADHD group and the control subjects, for both male and females, was TR14 (Table 1).
Discussion
Due to the major rise in reported ADHD prevalence during the past decades [38], the public debate concerning the need for more objective ADHD diagnosis has been vivid [19,39,40]. We have recently published an article with results showing specific ABR differences in the young female group with ADHD compared to healthy controls [14].
The aim of this study was to identify differences in brainstem responses, ABR, in young patients diagnosed with ADHD compared to a control group. To our knowledge this is the first ABR study done on young patients (7-18 years) with ADHD trying to find gender specific ABR differences. In this study we compared the young females with ADHD both to a young female control group but also to young males with ADHD and their male control group. In the group of young males with ADHD we found three traits specific for ADHD compared to healthy controls; TR4, TR5 and TR14. In the young female group we found three traits not present in the healthy controls; TR6, TR14 and TR15. One trait co-occurred in both gender groups; TR14.
Looking at the ABR findings from a biological view, two of the female traits (TR6 and TR14) and one of the male traits (TR4) were found in the thalamic area or in the near proximity. This supports other studies that have shown structural and functional abnormalities in the deep parts of the brain of young patients with ADHD [41-43]. It has been described earlier that thalamic dysfunction leads to symptoms of hyperactivity, inattention and dysregulation of sleep and wakefulness all of which are symptoms of ADHD [44,45]. The only trait that is not in the thalamic area is TR5 representing the cochlear nucleus, a more peripheral part of the brain. The cochlear nucleus is where parts of the auditory temporal processing occurs. It is an important component in sound detection in noisy environments and in selective auditory attention [46], deficits in these aspects of sound processing are frequent symptoms in ADHD.
TR15, only present in the young female ADHD group is located in lateral leminiscus; pons area. Previous studies regarding brain activity have shown higher resting-state activity in young patients with ADHD compared to controls in this area [47].
TR14 which is present in both gender groups is located in the midbrain ranging from SOC that is the first major site of convergence of auditory information from both ears important for localization of sound to thalamus. In Thalamus, transformation of soundsource acoustics (frequency, time and amplitude) into perceptual features (i.e. acoustic features) begins to form [48].
It is possible that these different traits found represent different subgroups of ADHD. Further research areas would be to link the person with a specific ABR trait to its reported symptoms. For example do patients with prominent TR5 report more ADHD symptoms involving hearing and sound detection?
Early studies have shown age and gender differences in ABR [49], whereas there are other more modern studies that demonstrate that the aging process is essentially a peripheral phenomenon which does not involve the central part of the acoustic pathways [50-52]. Neurodevelopmental differences may, at least partially, explain why girls are diagnosed so much later than boys [53]. Since our patients are age matched we believe that we have fully controlled for this factor in our study.
It is clear from our material that ABR in young patients with ADHD is gender specific. Only one trait namely TR14 overlapped; the others are gender specific.
Our findings support the fact that ADHD needs to be looked upon as a diagnosis composed of subgroups [9-12,54,55] a view that DSM5 and its subgrouping of ADHD supports. Our study shows that ABR has the potential to be the objective diagnostic instrument child and adolescent psychiatry has been looking for. ABR is a non-invasive method that is easily administered to patients; it is not dependent on language skills or cultural background [19]. Our findings suggest that ABR could be used as a complement and a support when diagnosing young patients with ADHD. By means of our ABR findings we have got a deeper understanding of the biological nature of ADHD and its subgroups. The present study suggests that the ABR method might provide useful biomarkers to support the clinical diagnoses of ADHD.
Many reports show possible regulation of locus coeruleus activity by metylphenidates and by atomoxetin [56-59]. Future research areas of interest would be to investigate whether medication would decrease the ABR differences between the group of children with ADHD and healthy controls, and if it is possible to predict choice of medical treatment according to trait match. There is also a need for longitudinal studies with child and adolescent control subjects with other psychiatric diagnoses to further substantiate our findings. We have therefor recently initiated a study of ABR and ASD in young patients. Another study regarding ABR and OCD has started in the autumn of 2015 at the University of Lund. Findings from these studies will be of great interest to investigate possible overlap with ADHD traits and the possible role of comorbidity.
Acknowledgments
This study was supported by grants from the local regional founds of Region Skåne. We would like to thank Edwin Klint Bywater for excellent statistical assistance. We would like to thank Barbro Knutsson for language review.
References
- Kieling R, Rohde LA (2012) ADHD in children and adults: diagnosis and prognosis. Curr Top BehavNeurosci 9: 1-16.
- American Psychiatric Association (2000) Diagnostic and statistical manual of mental disorders (4th ed) American Psychiatric Association, Washington, DC.
- American Psychiatric Association (2013) Diagnostic and Statistical Manual of Mental Disorders (5th ed) American Psychiatric Association, Arlington, VA.
- FaraoneSV, Biederman J, Mick E (2006)The age-dependent decline of attention deficit hyperactivity disorder: a meta-analysis of follow-up studies. Psychol Med 36: 159-65.
- Polanczyk G, Rohde LA (2007) Epidemiology of attention-deficit/hyperactivity disorder across the lifespan. CurrOpin Psychiatry 20: 386-392.
- PerouR, Bitsko RH, Blumberg SJ, Pastor P, Ghandour RM, et al. (2013) Mental health surveillance among children--United States, 2005-2011. MMWR. SurveillSumm 62: 1-35.
- Froehlich TE, Lanphear BP, Epstein JN, Barbaresi WJ, Katusic SK, et al. (2007) Prevalence, recognition, and treatment of attention-deficit/hyperactivity disorder in a national sample of US children. Arch PediatrAdolesc Med 161: 857-864.
- Nøvik TS, Hervas A, Ralston SJ, Dalsgaard S, Rodrigues PereiraR, et al. (2006) Influence of gender on attention-deficit/hyperactivity disorder in Europe--ADORE. Eur. Child Adolesc. Psychiatry; 15: I15-I24.
- Barkley RA, DuPaul GJ, McMurray MB (1990) Comprehensive evaluation of attention deficit disorder with and without hyperactivity as defined by research criteria. J Consult ClinPsychol 58: 775-789.
- Newcorn JH, Halperin JM, Jensen PS, Abikoff HB, Arnold LE, et al. (2001) Symptom profiles in children with ADHD: effects of comorbidity and gender. J Am Acad Child Adolesc Psychiatry 40: 137-146.
- Biederman J, Mick E, Faraone SV, Braaten E, Doyle A, et al. (2002) Influence of gender on attention deficit hyperactivity disorder in children referred to a psychiatric clinic. Am J Psychiatry 159: 36-42.
- Biederman J, Faraone SV (2004) The Massachusetts General Hospital studies of gender influences on attention-deficit/hyperactivity disorder in youth and relatives. Psychiatry Clinics of North America 27: 225-232.
- Gau SS, Shang CY (2010) Executive functions as endophenotypes in ADHD: evidence from the Cambridge Neuropsychological Test Battery (CANTAB). J Child Psychol Psychiatry 51: 838-849.
- Claesdotter-Hybbinette E, Safdarzadeh-Haghighi M, Råstam M, Lindvall M (2015) Abnormal brainstem auditory response in young females with ADHD. Psychiatry Res 229: 750-754.
- Hirsch O, Christiansen H (2015) Faking ADHD? Symptom Validity Testing and Its Relation to Self-Reported, Observer-Reported Symptoms, and Neuropsychological Measures of Attention in Adults. J AttenDisord 5.
- Hult N, Kadesjö J, Kadesjö B, Gillberg C, Billstedt E (2015) ADHD and the QbTest: Diagnostic Validity of QbTest. J AttenDisord.
- Rajendran K, O'Neill S, Marks DJ, Halperin JM (2015) Latent profile analysis of neuropsychological measures to determine preschoolers' risk for ADHD. J Child Psychol Psychiatry 56: 958-965.
- Jewett DL, Williston JS (1971) Auditory-evoked far fields averaged from the scalp of humans. Brain 94: 681-696.
- Bywater EK (2014) Features of Auditory Brainstem Response SpectralRepresentations as Tools for ADHD-Diagnostics. Lund´s University.
- Sand T (1990) Statistical properties of ABR amplitudes and latencies. Implications for computation of reference limits and relation to click phase. ScandAudiol 19: 131-137.
- Musiek FE, Lee WW (1995) The auditory brain stem response in patients with brain stem or cochlear pathology. Ear Hear 16: 631-636.
- O'Connor K (2012) Auditory processing in autism spectrum disorder: a review. NeurosciBiobehav Rev 36: 836-854.
- Taylor MJ, Rosenblatt B, Linschoten L (1982) Auditory brainstem response abnormalities in autistic children. Can J NeurolSci 9: 429-433.
- Wong V, Wong SN (1991) Brainstem auditory evoked potential study in children with autistic disorder. J Autism DevDisord 21: 329-340.
- Maziade M, Mérette C, Cayer M, Roy MA, Szatmari P, et al. (2000) Prolongation of brainstem auditory-evoked responses in autistic probands and their unaffected relatives. Arch Gen Psychiatry 57: 1077-1083.
- KwonS, Kim J, Choe BH, Ko C, Park S (2007)Electrophysiologic assessment of central auditory processing by auditory brainstem responses in children with autism spectrum disorders. J Korean Med Sci 22: 656-659.
- Hitoglou M, Ververi A, Antoniadis A, Zafeiriou DI (2010) Childhood autism and auditory system abnormalities. PediatrNeurol 42: 309-314.
- Källstrand J, Olsson O, Nehlstedt SF, Sköld ML, Nielzén S (2010) Abnormal auditory forward masking pattern in the brainstem response of individuals with Asperger syndrome. Neuropsych Dis and Treat 24: 289-296.
- Baghdassarian E, Källstrand J, Nielzén S, Lewander T (2014) P.1.c.011Brainstem evoked response audiometry biomarkers in patients with schizophrenia and adult ADHD. EuroNeuropsychopharmaco 24: 187-188
- JohnstonBA, Mwangi B, Matthews K, Coghill D, Konrad Steele JD (2014) Brainstem abnormalities in attention deficit hyperactivity disorder support high accuracy individual diagnostic classification. Hum Brain Mapp 35: 5179-5189.
- Källstrand J, Nehlstedt SF, Sköld ML, Nielzén S (2012) Lateral asymmetry and reduced forward masking effect in early brainstem auditory evoked responses in schizophrenia. Psychiatry Res 196: 188-193.
- Nielzén S, Olsson S, Källstrand J, Nehlstedt S (2008) Aberrant brain stem function in schizophrenia. European Psychiatry 23: 135-137.
- Sköld M,Källstrand J,NehlstedtS, Nordin A, NielzénS, et al.(2014)Thalamocortical abnormalities in auditory brainstem response patterns distinguish DSM-IV bipolar disorder type I from schizophrenia. J Affective Disorders 169: 105-111.
- Ismail N, Amin A (1999) Auditory Brainstem Response in Attention Deficit Hyperactivity Disorders in Children. Current Psychiatry 6: 63-70.
- Schochat E, Scheuer CI, Andrade ER (2002) ABR and auditory P300 findings in children with ADHD. ArqNeuropsiquiatr 60: 742-747.
- Klin A (1993)Auditory brainstem responses in autism: brainstem dysfunction or peripheral hearing loss? Journal of Autism and Developmental Disorder. 23: 15-35.
- Parkkonen L, Fujiki N, Mäkelä JP (2009) Sources of auditory brainstem responses revisited: contribution by magnetoencephalography. Hum Brain Mapp 30: 1772-82.
- Beck M (2010) Mind Games: Attention-Deficit Disorder Isn’t Just for Kids. Why Adults Are Now Being Diagnosed, Too.
- GregoireC (2014) Worldwide ADHD Rates Are Higher Than Ever, And It Might Be America's Fault.
- Rettew D (2014)The ADHD Debate. Psychology Today Online, January 6, ABCs of Child Psychiatry.
- Dickstein SG, Bannon K, Castellanos FX, Milham MP (2006) The neural correlates of attention deficit hyperactivity disorder: an ALE meta-analysis. J Child Psychol Psychiatry 47: 1051-1062.
- Zhu CZ, Zang YF, Cao QJ, Yan CG, He Y, et al. (2008) Fisher discriminative analysis of resting-state brain function for attention-deficit/hyperactivity disorder Neuroimage 40: 110-120.
- Cortese S, Imperati D, Zhou J, Proal E, Klein RG, et al. (2013) White matter alterations at 33-year follow-up in adults with childhood attention-deficit/hyperactivity disorder Biol Psychiatry 74: 591-598.
- Steriade M, Llinás RR (1988) The functional states of the thalamus and the associated neuronal interplay Physiol Rev 68: 649-742.
- Ivanov I, Bansal R, Hao X, Zhu H, Kellendonk C, et al.(2010) Morphological abnormalities of the thalamus in youths with attention deficit hyperactivity disorder Am J Psychiatry 167: 397-408.
- Fortune T, Lurie DI (2009) Chronic low-level lead exposure affects the monoaminergic system in the mouse superior olivary complex J Comp Neurol 513: 542-558.
- Zang YF, He Y, Zhu CZ, Cao QJ, Sui MQ, et al. (2007) Altered baseline brain activity in children with ADHD revealed by resting-state functional MRI Brain Dev 29: 83-91
- Bartlett EL (2013) The organization and physiology of the auditory thalamus and its role in processing acoustic features important for speech perception Brain Lang 126: 29-48.
- Allison T, Wood CC, Goff WR (1983) Brainstem auditory, pattern-reversal visual, and short-latency somatosensory evoked potentials: latencies in relation to age, sex, and brain and body size ElectroencephalogrClinNeurophysiol 55: 619-36.
- Costa P, Benna P, Bianco C, Ferrero P, Bergamasco B (1990) Aging effects on brainstem auditory evoked potentials ElectromyogrClinNeurophysiol30: 495-500.
- Burkard RF, Sims D (2001)The human auditory brainstem response to high click rates: aging effects Am J Audiol 10: 53-61.
- Konrad-Martin D, Dille MF, McMillan G, Griest S, McDermott D, et al. (2012) Age-related changes in the auditory brainstem response J Am AcadAudiol 23: 18-35.
- Mahone EM, Wodka EL (2008) The neurobiological profile of girls with ADHD DevDisabil Res Rev 14: 276-284.
- Graetz BW, Sawyer MG, Baghurst P (2005) Gender differences among children with DSM-IV ADHD in Australia J Am Acad Child Adolesc Psychiatry 44: 159-168.
- Levy F, Hay DA, Bennett KS, McStephen M (2005) Gender differences in ADHD subtype comorbidity J Am Acad Child Adolesc Psychiatry 44: 368-376.
- Harro J, Meriküla A, Lepiku M, Modiri AR, Rinken A, et al.(2000) Lesions of locus coeruleus projections by DSP-4 neurotoxin treatment: effect on amphetamine-induced hyperlocomotion and dopamine D2 receptor binding in rats. PharmacolToxicol 86: 197-202.
- Devilbiss DM,Berridge CW (2006) Low-dose metylphenidate actions on tonic and phasic locus coeruleus discharge J PharmacolExpTher 319: 1327-1335.
- Howells FM, Stein DJ, Russell VA (2012) Synergistic tonic and phasic activity of the locus coeruleus norepinephrine (LC-NE) arousal system is required for optimal attention performance Metab Brain Dis 27: 267-274.
- Bari A, Aston-Jones G (2013)Atomoxetin modulates spontaneous and sensory-evoked discharge of locus coeruleus noradrenergic neurons Neuropharmaco logy 64: 53-64.