Research Article - (2018) Volume 8, Issue 4
The Use of Short Form 36 and Beck Depression Inventory in Acute Cervical Spinal Cord Injury Patients
- Corresponding Author:
- Hung-Chen Wang
Department of Neurosurgery, Kaohsiung Chang Gung Memorial Hospital 123, Ta Pei Road, Niao Sung Dist., Kaohsiung, Taiwan
Tel: +886-7-7317123
Fax: +886-7-731-8762
Abstract
Objective
Spinal cord injury (SCI) is one of the major catastrophes in life. After injury, most patients suffer from varying degrees of physical disabilities as well as psychological impacts, resulting in lower quality of life. The objective of this study is to evaluate the physical and mental impacts in patients with acute SCI.
Methods
A prospective trial to evaluate the use of short form 36 (SF-36) and Beck Depression Inventory (BDI) in acute cervical SCI patients. A total of 40 patients with acute SCI were included in the study. SF-36, BDI, the Hopkins Rehabilitation Engagement Rating Scale and Japanese Orthopedic Association Score of individual patients were recorded at three points -- within two weeks and one month, three months, and six months after injury.
Results
Patients with acute SCI had significantly poorer levels of SF-36 domains, such as PF, RP, BP, SF and RE, PCS and BDI scores compared with controls. SF-36 score was positively correlated with JOA score on PF, RP, GH, and PCS, but negatively correlated with BDI on all domains components except BP. The excellent JOA recovery group had higher SF-36, lower BDI score on all time points then non-excellent group. However, compared to the HRERS, there was only statistically significant higher score during the first month of the excellent recovery group.
Conclusions
BDI and SF-36 are good assessment tools for evaluating the physiological and psychological status in patients with SCI. They can help health care givers to understand the respective statuses of the SCI patients to improve their quality of life.
Keywords
Spinal cord injury, Short form 36, Beck depression inventory, Japanese orthopedic association score, The hopkins rehabilitation engagement rating scale
Introduction
Spinal cord injury (SCI) occurs when the spinal cord is severely bruised, compressed, lacerated, or severed as a result of traumatic injury or disease. SCI is usually highly debilitating, resulting in the loss of sensation, muscle paralysis, and autonomic dysregulation that are capable of impacting the functioning of multiple body systems. SCI patients, therefore, often require ongoing medical and ancillary treatment. SCI is also associated with the development of secondary conditions such as chronic pain, infections, and chronic fatigue, all contributing to lower quality of life (QoL) and potentially reduced social participation [1-4]. There is convincing evidence that the prevalence of mental health disorders, such as depression, anxiety and post-traumatic stress syndrome, are elevated in persons with chronic SCI [5].
Regardless of the current progress in medical sciences and technology, it is still too easy to cause permanent neurological injury even after the appropriate surgery or pharmaceutical treatment. Most SCI patients are still left with different degrees of limb dysfunction and complications after all the necessary treatments [6]. At the psychological level, some patients become closed and socially isolated, prone to guilt, low self-esteem, low self-worth, and exhibit melancholy symptoms [7,8]. For patients that showed depressive symptoms, and yet not treated in time, the ones with severe pessimism could lead to a higher level of suicidal ideation compare to the average person, thus causing great regret in the family and society [9-11]. Therefore, raising the quality of life of SCI patients has gradually become one of the main objectives of the rehabilitation program for spinal cord injury and the evaluation of the outcome of rehabilitation program [12,13]. Patients with spinal cord injury were given proper disposal during acute period to minimize their damage. In order to achieve individual patient’s ability to restore their independence, we should grasp the opportunity to elevate functioning of the limbs to their best condition, while avoiding the negative sequelae, in the later period. We can improve the emphasis of rehabilitation to self-care ability as the main purpose, and give support to social resources, and improve the QoL of spinal cord injury.
The definition of the term “quality of life” may differ from one person to another, based on their feelings and cognitions. McKeon considers the concept of quality of life as Aristotle, who argues that happiness represents the relevant theory of a good Life [14]. Until the 1960s, the concept of quality of life was widely used in the study of Health [15]. In the study of the quality of life of spinal cord injury, there are both a subjective and an objective perspective. On the one hand, the subjective method defines quality of life based on personal subjective feeling of hope and achievement consistent with the subjects. On the other hand, the objective method is to predict the subjective quality of life by assessing the objective damages of each case. Therefore, scholars believe that subjective feelings can be used to directly assess the quality of life, whereas the objective quality of life measurement can only describe the living conditions and experience, but not as a direct measure for the quality of life [1,2,7-9,12-14].
The purpose of this study was to: (1) to understand the depression and QoL of patients with acute cervical SCI; (2) To explore the relationship between depression and QoL in patients with acute cervical SCI; and (3) to explore whether the predictors of depression and QoL affecting the acute cervical SCI were important predictors of recovery in JOA score.
Methods
▪ Patients
The method of diagnosis for acute cervical SCI was based on clinical symptoms and spinal imaging, including X-ray, computed tomography (CT) and magnetic resonance imaging (MRI). Patients were excluded if (1) they had previous cervical spine trauma; (2) the duration between injury and admission to our hospital was more than 24 hours; (3) they had severe underlying diseases; or (4) had severe multiple trauma with unstable hemodynamic status.
Twenty patients were excluded from this study, including eight who refused to participate, two with previous cervical trauma, two with severe underlying diseases (one end stage renal failure and one severe liver cirrhosis) and eight with cervical SCI more than 24 h. In total, 40 adult patients who had suffered from acute blunt cervical SCI were included in the study. The mechanisms of injury included 29 traffic accidents, 10 falling accidents, and 1 collision with heavy objects. The Ethics Committee of the hospital’s Institutional Review Board (Chang Gung Memorial Hospital Ethics Committee) Approved the study (103- 5218B and 106-1104C). All of the patients or their representatives provided written informed consent. For comparison, 40 sex- and age-matched volunteers were recruited as controls. All necessary written informed consents from any healthy volunteers involved in the study was provided, including consents to participate in the study where appropriate.
The scale of questionnaires for this study was filled-in by the subject-in-case or by the researcher after completing the explanation of the language, followed by the use of Beck Depression Inventory (BDI) and SF-36 quality of life questionnaire. The Hopkins rehabilitation engagement rating scale (HRERS) score was used to conduct a questionnaire survey at three points, within two week and one month, three months and six months after injury.
Short Form-36 (SF-36) is a 149 health questionnaire from medical outcome Study by Dr. John Ware, Jr, developed in 1985, since the International Association for the Quality of life assessment (International Quality Assessment, Iqola). In 1991, with the SF-36 scale as a tool for measuring the quality of life. The SF-36 scale is a multidimensional scale to measure the quality of life that is applicable to all ages, diseases and treatments. The scale is divided into eight operational measurements of the health of the surface, each of the construction of a number of topics to evaluate, including: body function (physical functioning, PF), Body Function- Role Limited (roles limitations of physical Problems, RP), body pain degree (bodily Pain, BP), overall health (General health, GH), energy status (vitality, VT), social function (Social functioning, SF), psychological function-role Limited (roles emotional, RE), mental Health (mental health, MH); These eight healthy functional facets can be converted to physical health (Physical component Score, PCS, and mental health (Mental Component score, MCS). Two, scores from 0 points to 100 points, the higher the score, indicating the better health function [16].
The Hopkins Rehabilitation Engagement Rating Scale (HRERS) is a 5-item, clinician-rated measure developed to quantify engagement in acute rehabilitation services [17]. The HRERS is a valid and reliable measure of rehabilitation engagement that relates to intermediate-term functional outcomes.
The BDI-II contains 21 questions, each answer being scored on a scale value of 0 to 3 [18]. Higher total scores indicate more severe depressive symptoms. The standardized cutoffs used here differ from the original as such: 0-13: minimal depression; 14–19: mild depression; 20–28: moderate depression; and 29–63: severe depression.
▪ Clinical manifestations
The neurological statuses of patients were recorded at the time of admission using the Japanese Orthopedic Association (JOA) cervical spine myelopathy functional assessment score [19]. The Abbreviated Injury Score (AIS) for individual body regions was determined and the total extent of the injury was calculated using the objective Injury Severity Score (ISS) upon admission [20]. The choices of operation depended on imaging findings and clinical presentations. The neurological status of each patient was evaluated at admission and at 6 months follow-up according to the JOA disability scale. The neurological recovery rate was calculated using the Hirabayashi method: (6 months follow-up JOA score – at admission score)/(18 - at admission score) x 100% [21]. Recovery rates were graded as follows: ≧ 75%, excellent; 50–74%, good; 25–49%, fair; and <25%, poor [22].
▪ Statistics
Category data was analyzed by means of the chi-square test or Fisher’s exact test. Continued data was analyzed using the Student’s t test or Mann- Whitney U test. Data are mean ± standard derivation (SD) for normally distributed data or median (inter-quartile range, IQR) for not-normally distributed data. The SPSS 18.0 (SPSS Inc., Chicago, IL, USA) software was used for all statistical analyses.
Results
Of the 40 patients with acute SCI, the majority (33/40, 82.5%) were male with a mean age of 45 years (range of 20–67 years), who were severely injured with a mean injury severity score (ISS) of 21, and JOA score with a mean of 7. Thirty-four had cervical spine surgeries including 20 emergency surgeries and 31 elective surgeries. Demographic data and comparison with healthy controls were listed on (Table 1).
| Parameters | Acute SCI (n=40) |
Controls (n=40) |
p value | 95% CI (lower, upper) |
|---|---|---|---|---|
| Age (y) | 44.95 ± 14.20 | 44.13 ± 14.55 | 0.798 | -5.57, 7.22 |
| Male | 33 | 33 | ||
| Body mass index | 25.97 ± 6.33 | 27.18 ± 6.87 | 0.867 | -6.43, 9.24 |
| Level of SCI | ||||
| C2-C4 | 31 | NA | ||
| C5-C7 | 9 | NA | ||
| Injury Severity Score at admission | 21.23 ± 13.88 | |||
| JOA score at admission | 6.98 ± 5.08 | NA | ||
| Neurosurgical intervention | 34 | NA | ||
| Short Form 36 | ||||
| Physical function | 21.47 ± 33.13 | 85.00 ± 11.21 | ≤ 0.001 | -75.56, -51.49 |
| Role physical | 13.97 ± 31.50 | 79.38 ± 21.84 | ≤ 0.001 | -77.82, -52.99 |
| Bodily pain | 46.33 ± 33.87 | 75.23 ± 12.61 | ≤ 0.001 | -41.48, -16.31 |
| General health | 62.42 ± 21.11 | 62.73 ± 11.86 | 0.942 | -8.59, 7.99 |
| Vitality | 55.76 ± 22.92 | 61.50 ± 10.87 | 0.193 | -14.50, 3.01 |
| Social functioning | 59.85 ± 34.05 | 76.88 ± 17.35 | 0.012 | -30.18, -3.87 |
| Role emotional | 36.36 ± 46.67 | 74.17 ± 37.35 | ≤ 0.001 | -57.89, -17.72 |
| Mental health | 66.06 ± 18.87 | 65.45 ± 13.59 | 0.873 | -6.98, 8.20 |
| Physical component summary | 13.96 ± 17.24 | 48.65 ± 7.24 | ≤ 0.001 | -41.18, -28.21 |
| Mental component summary | 44.11 ± 14.49 | 49.10 ± 8.49 | 0.087 | -10.73, 0.74 |
| Beck Depression Inventory | 23.73 ± 10.74 | 6.67 ± 5.82 | ≤ 0.001 | |
| HRERS | 20.56 ± 6.40 | NA | ||
| Short Form 36, Beck Depression Inventory and HRERS were recorded between two weeks and one month after spinal cord injury. Abbreviations: SCI: Spinal Cord Injury; CI: Confidence Interval; JOA: Japanese Orthopaedic Association; HRERS: The Hopkins Rehabilitation Engagement Rating Scale; NA: Not Available. |
||||
Table 1: Demographic Data of Patients and Controls at Admission.
The mean eight domains score of SF-36 in patients with acute SCI, including PF, RE, BP, GH, VT, SF, RE and MH were 21.47 ± 33.13, 13.97 ± 31.50, 46.33 ± 33.87, 62.42 ± 21.11, 55.76 ± 22.92, 59.85 ± 34.05, 36.36 ± 46.67 and 66.06 ± 18.87, respectively (Table 1). The mean two components score of SF-36 in patients with acute SCI, including PCS and MCS were 13.96 ± 17.24 and 44.11 ± 14.49, respectively. Patients with acute SCI had significantly poorer levels of SF-36 domains, including PF, RP, BP, SF and RE, physical component and BDI scores compared with the controls (p ≦ 0.001, p ≦ 0.001, p ≦ 0.001, p=0.012, p ≦ 0.001, and p ≦ 0.001, respectively) (Table 1).
▪ Effects of risk factors on each domain and component summary of SF-36
A correlation analysis was used to test the influence of age, body mass index, ISS, JOA score, BDI score and HRERS on each domain and component summary of SF-36 (Table 2). Based on the statistical analyses (correlation coefficient, p value), domains and component summary of SF-36 score were positively correlated with JOA score on PF, RP, GH, and PCS, but negatively correlated with BDI on all domains components except for BP (Table 2).
| Variables | Age | Body Mass Index | ISS | JOA score | BDI | HRERS | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| r | P value | r | P value | r | P value | r | P value | r | P value | r | P value | |
| Physical function | -0.258 | 0.14 | -0.019 | 0.916 | -0.27 | 0.129 | .690** | ≤ 0.001 | -.545** | 0.001 | 0.299 | 0.097 |
| Role physical | -0.083 | 0.64 | -0.088 | 0.628 | -0.202 | 0.258 | .494** | 0.003 | -.460** | 0.006 | 0.29 | 0.107 |
| Bodily pain | 0.003 | 0.987 | -0.019 | 0.916 | 0.005 | 0.978 | -0.026 | 0.884 | -0.217 | 0.226 | 0.109 | 0.558 |
| General health | -0.137 | 0.446 | -0.102 | 0.58 | -0.281 | 0.119 | .430* | 0.012 | -.448** | 0.009 | 0.222 | 0.229 |
| Vitality | -0.207 | 0.247 | 0.061 | 0.739 | -0.112 | 0.543 | 0.16 | 0.374 | -.436* | 0.011 | 0.141 | 0.45 |
| Social functioning | -0.136 | 0.451 | -0.1 | 0.588 | 0.061 | 0.74 | 0.208 | 0.245 | -.431* | 0.012 | 0.1 | 0.591 |
| Role emotional | -0.291 | 0.101 | 0.118 | 0.519 | -0.056 | 0.761 | 0.34 | 0.053 | -.382* | 0.028 | 0.033 | 0.861 |
| Mental health | -.475** | 0.005 | 0.17 | 0.353 | -0.332 | 0.064 | 0.32 | 0.07 | -.473** | 0.005 | 0.083 | 0.657 |
| Physical component summary | -0.146 | 0.417 | -0.104 | 0.57 | -0.225 | 0.217 | .544** | 0.001 | -.604** | ≤ 0.001 | 0.223 | 0.227 |
| Mental component summary | -.347* | 0.048 | 0.153 | 0.402 | -0.189 | 0.301 | 0.318 | 0.071 | -.571** | 0.001 | 0.1 | 0.593 |
| r: correlation coefficient. * indicates that p value <0.05. ** indicates that p value <0.01. Short Form 36, Beck Depression Inventory and HRERS were recorded between two weeks and one month after spinal cord injury. ISS and JOA score were recorded at the time of admission. Abbreviations: ISS: Injury Severity Score; JOA: Japanese Orthopaedic Association; BDI: Beck Depression Inventory; HRERS: The Hopkins Rehabilitation Engagement Rating Scale. |
||||||||||||
Table 2: Coefficients analysis of component summary score and each domain of Short Form 36 during hospitalization in patients with acute spinal cord injury.
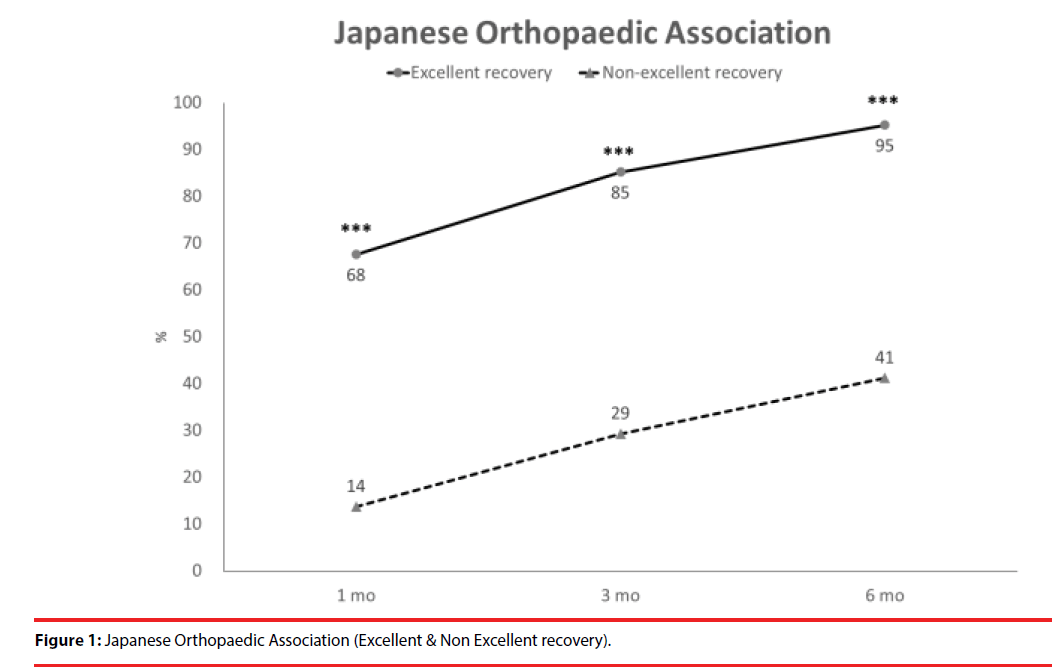
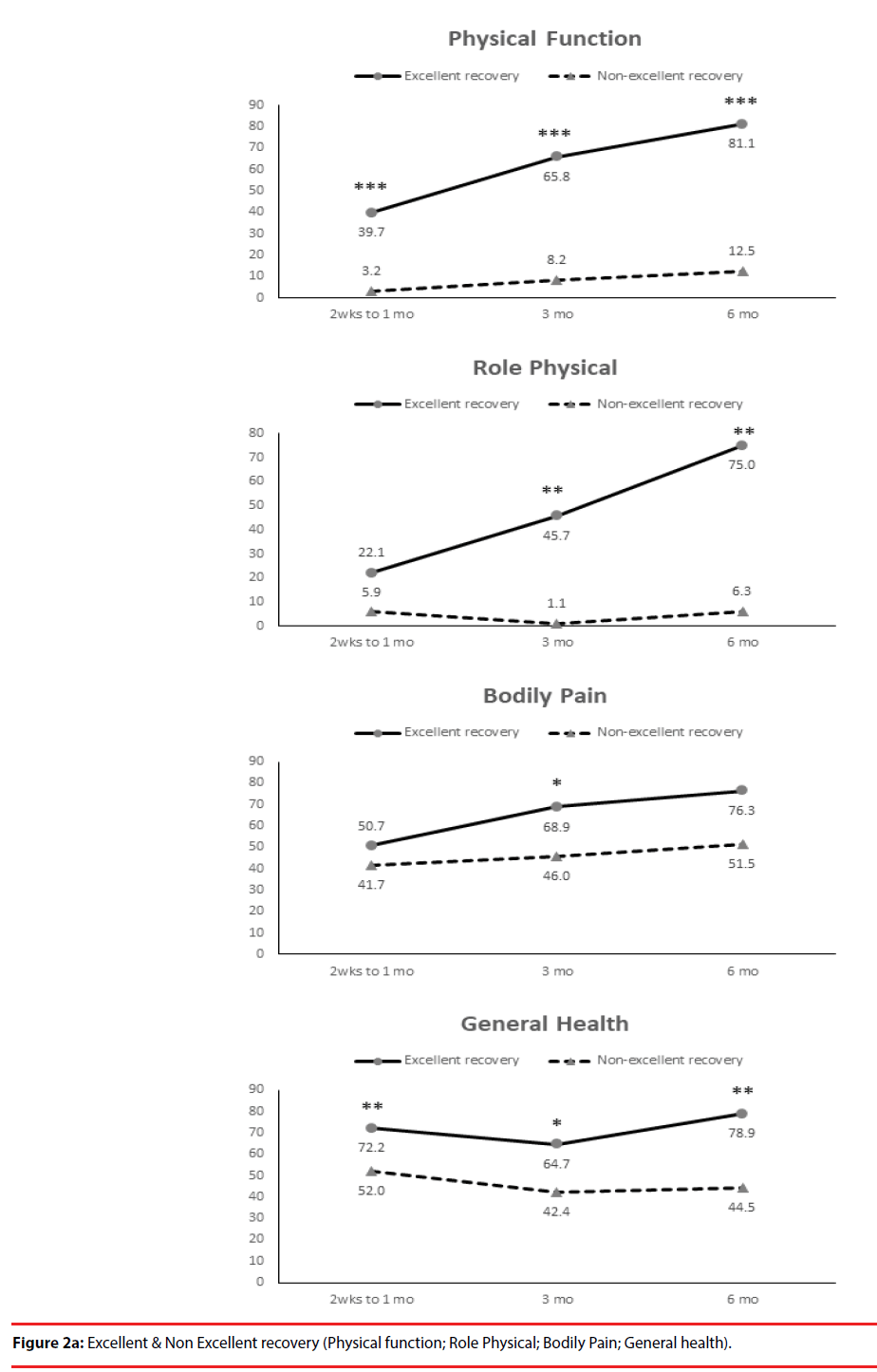
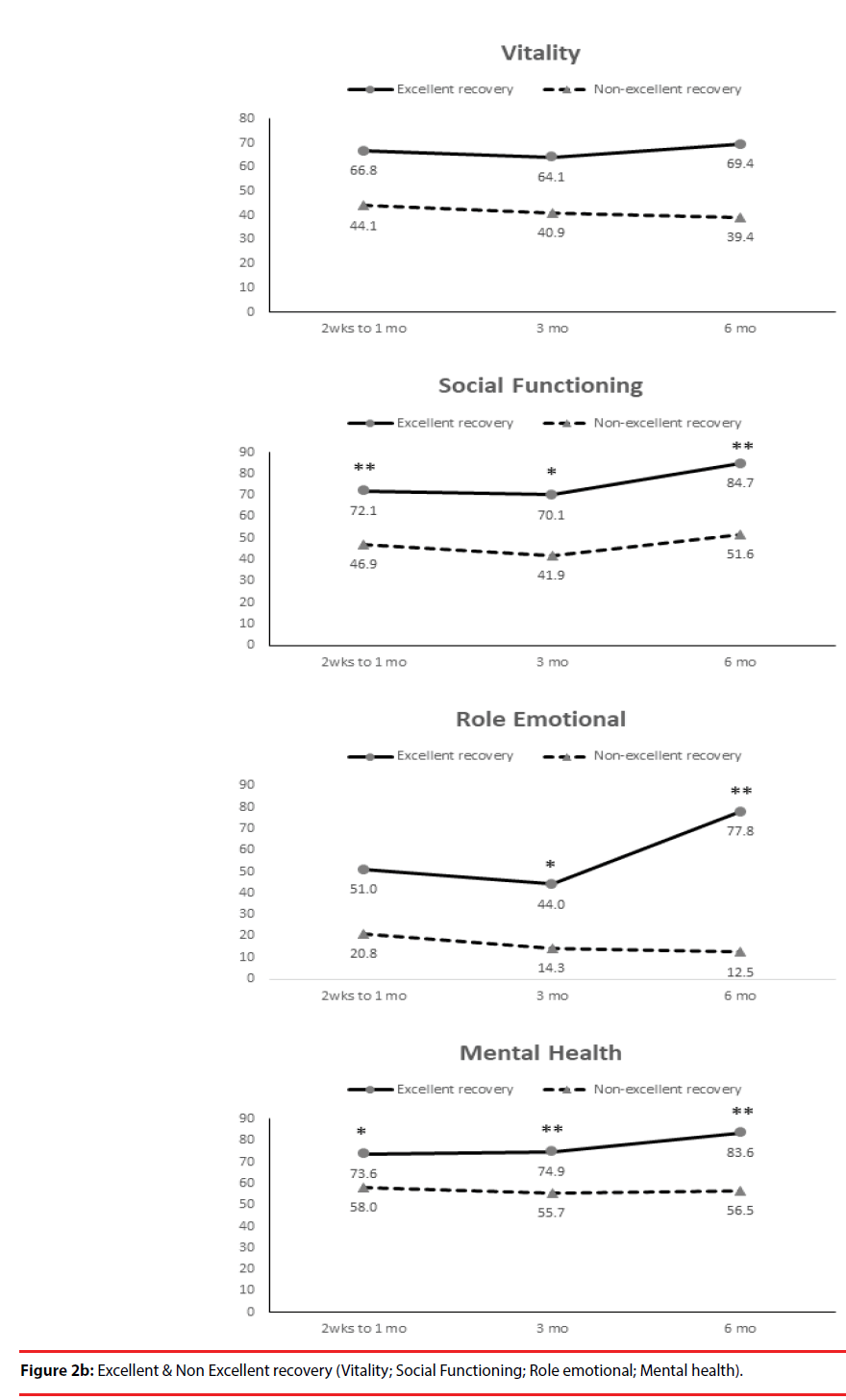
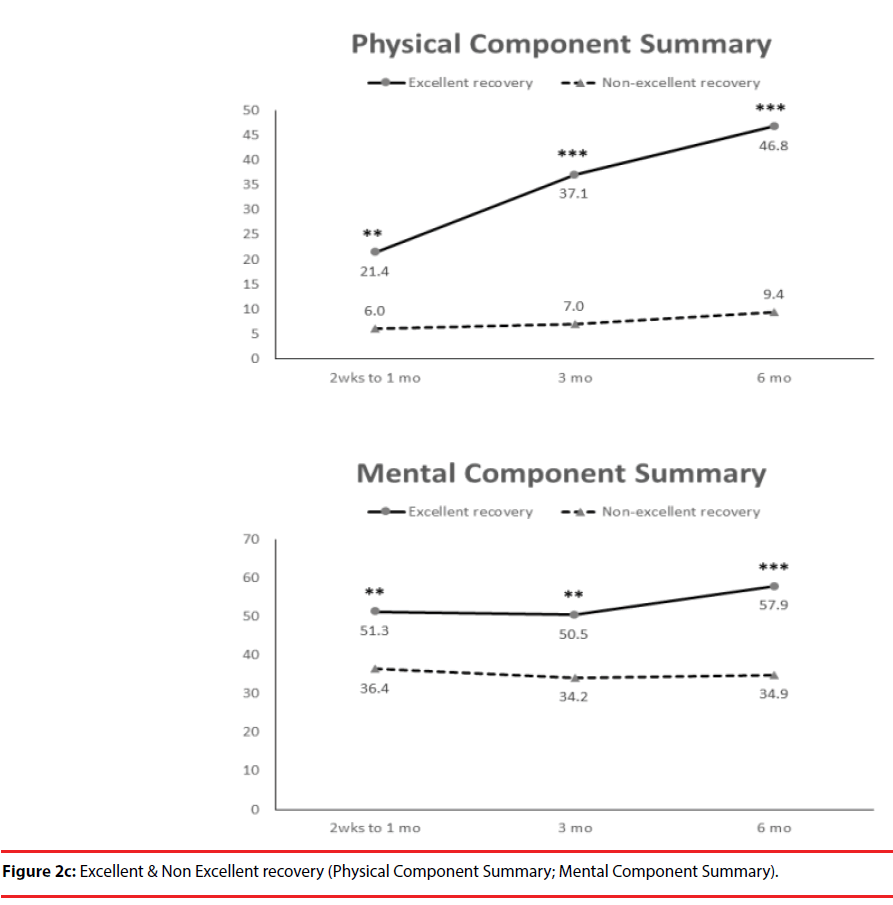
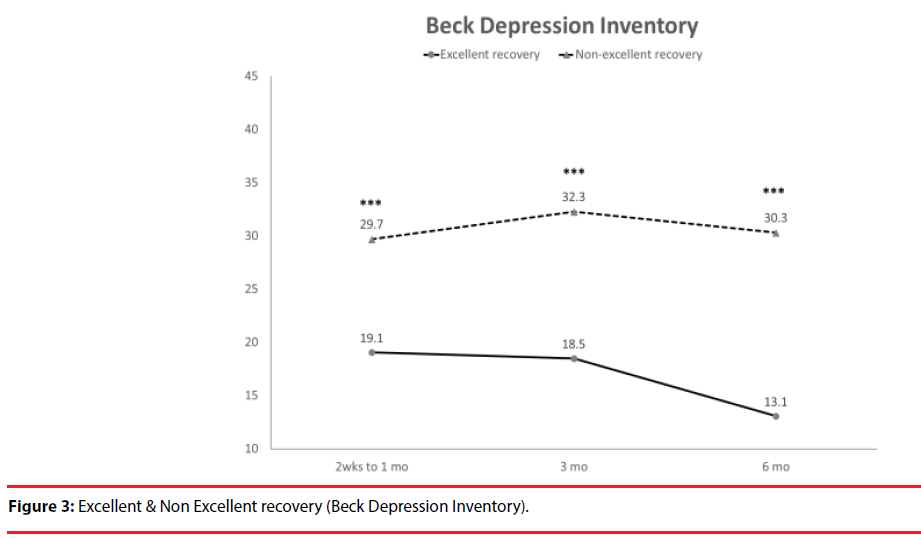
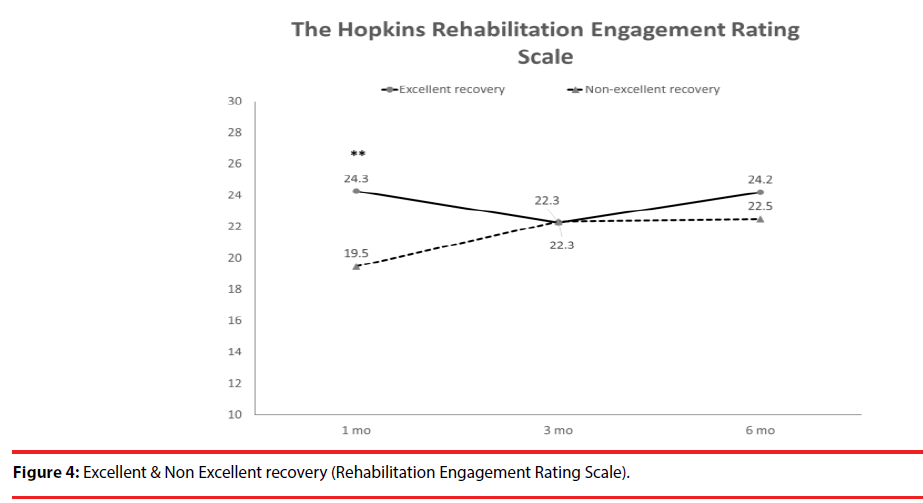
According the results of JOA recovery at six months-post-trauma, we divided our patients into two groups. Patients with JOA recovery more than 75% was defined as excellent outcome and less than 75% was defined as non-excellent recovery (Figure 1). Compared to patients who had excellent recovery and had non-excellent recovery at 6 month post trauma, there were statistically significant differences in JOA score at admission (p=0.012), BDI score within one month (p=0.001) and HRERS within one month (p=0.006), but no statistically significant difference in age (p=0.072), body mass index (p=0.689), level of SCI (p=0.687), ISS (p=0.060), and neurosurgical intervention (p=0.073) between the two groups (Table 3). Compared to the SF-36 scores between the two group, the excellent recovery group had higher SF-36 than non-excellent recovery group from hospitalization to 6 month follow up. There were statistically significant differences on all domains (except RP, BP and RE during hospitalization, and VT on all time points) and two components summary (Figures 2A-2C). The excellent recovery group had significantly lower BDI score on all time points compared to the non-excellent group (Figure 3). However, compared to the HRERS, a significantly higher score was only observed during the first month in the excellent recovery group (Figure 4).
| Parameters | Excellent recovery (n=18) |
Non-excellent recovery (n=22) |
p value | Odds Ratio | 95% CI (lower, upper) |
|---|---|---|---|---|---|
| Age (y) | 39.0613.49 | 48.06 ± 14.69 | 0.072 | -18.84, 0.85 | |
| Male | 14 | 19 | 0.680 | 0.744 | 0.30, 1.84 |
| Body mass index | 25.09 ± 4.08 | 25.94 ± 7.66 | 0.685 | -5.11, 3.41 | |
| Level of SCI | |||||
| C2-C4 | 14 | 17 | 0.687 | 0.782 | 0.13, 1.94 |
| C5-C7 | 4 | 5 | |||
| Injury Severity Score at admission | 16.94 ± 3.92 | 24.90 ± 17.95 | 0.06 | -16.30, 0.38 | |
| JOA score at admission | 9.44 ± 4.27 | 4.89 ± 5.52 | 0.012 | 1.07, 8.03 | |
| JOA score at 6 months | 17.31 ± 1.11 | 9.31 ± 4.03 | ≤ 0.001 | 5.51, 10.49 | |
| JOA recovery rate at 6 months | 95.25 ± 8.45 | 41.23 ± 21.26 | ≤ 0.001 | 40.64, 67.40 | |
| Neurosurgical intervention | 13 | 21 | 0.073 | 0.270 | 0.04, 1.65 |
| Short Form 36 | |||||
| Physical function | 42.19 ± 36.88 | 3.67 ± 14.20 | ≤ 0.001 | 17.80, 59.24 | |
| Role physical | 23.44 ± 37.05 | 6.67 ± 25.82 | 0.153 | -6.65, 40.19 | |
| Bodily pain | 53.13 ± 34.78 | 36.86 ± 31.00 | 0.19 | -8.53, 41.07 | |
| General health | 71.75 ± 20.39 | 49.79 ± 17.58 | 0.004 | 7.62, 36.31 | |
| Vitality | 64.69 ± 19.87 | 42.14 ± 17.62 | 0.003 | 8.41, 36.68 | |
| Social functioning | 73.44 ± 31.25 | 41.96 ± 32.75 | 0.012 | 7.52, 55.43 | |
| Role emotional | 47.92 ± 48.64 | 23.81 ± 42.22 | 0.161 | -10.20, 58.42 | |
| Mental health | 72.00 ± 16.59 | 58.29 ± 18.61 | 0.042 | 0.55, 26.88 | |
| Physical component summary | 22.89 ± 19.04 | 5.45 ± 10.57 | 0.004 | 6.01, 28.86 | |
| Mental component summary | 50.33 ± 12.13 | 35.59 ± 13.20 | 0.004 | 5.27, 24.21 | |
| Beck Depression Inventory | 18.81 ± 7.37 | 30.11 ± 9.76 | 0.001 | -17.40, -5.19 | |
| HRERS | 23.00 ± 6.82 | 19.61 ± 5.18 | 0.006 | 1.45, 8.14 | |
| Short Form 36, Beck Depression Inventory and HRERS were recorded between two weeks and one month after spinal cord injury. ISS and JOA score were recorded at the time of admission. Abbreviations: ISS: Injury Severity Score; JOA: Japanese Orthopaedic Association; BDI: Beck Depression Inventory; HRERS: The Hopkins Rehabilitation Engagement Rating Scale. |
|||||
Table 3: Factors associated with spinal cord injury recovery.
Discussion
The SF-36 questionnaire is a valid, reliable and concise measure of health that is useful for the evaluation of the efficacy of interventions, as well as for determining the relative burdens of various diseases [23-25]. In the present study, patients with acute SCI had significantly lower levels of SF-36 domains, including PF, RP, BP, SF and RE, physical component summary and BDI scores compared with the controls, but not in their mental component summary (Table 1). We compared the SF-36 with the standard BDI questionnaire, and we found that the SF-36 were negatively correlated with BDI on all domains components, except for BP, at a statistically significant level (Table 2).
Depression was highly prevalent in individuals with SCI and was related to some demographic, pathophysiological, and socioeconomic indicators. The primary predictive indicators and the factors influencing depression should be determined in order to provide early detection and timely treatment for preventing more complications as well as improving the quality of life for individuals with SCI. From a meta-analysis, the mean prevalence estimate of depression diagnosis after SCI was 22.2% indicating that the prevalence of depression after SCI is substantially greater than that in the general medical population [26]. Previous studies also showed elevated levels of depressive mood states among the chronic SCI population, with an upward rate ranging between 19% and 30% [26-29]. A recent study by Khazaeipour et al. reported that up to 49.3% (66/134) of patients with SCI had mild to severe depression, according to their BDI scores [30]. A systematic review showed that longitudinal studies revealed that symptoms of depression had continual impacts on psychosocial outcomes up to 10 years post-injury [31]. Longitudinal examination of the levels of anxiety and depression among patients after spinal cord injury, Kennedy et al. data showed BDI scores were the highest during the acute phase of the injury and at the months leading up to discharge [29]. In congruent with the previous study, our data demonstrated a significantly higher BDI scores in patients with non-excellent recovery compared with patient with excellent JOA recovery from acute phase of the injury and persisted to 6 months. Furthermore, patients with excellent JOA recovery had significantly decreased BDI scores at 6 month compared to the first month and at 3 month (p=0.009 and p=0.008, respectively) (Figure 3).
Our results showed that only the bodily pain domain was not significant correlated with BDI during acute phase. Our results were consistent with other studies that showed depressive symptoms and BDI scores were not associated with pain severity [32-34]. Tate et al. reported that age was related to pain, with younger subjects reporting higher levels [33]. However, when SCI patients with chronic pain had higher depression ratings and their BDI scores were correlated with some of the SF-36 domains (general health, vitality, social functioning and mental health) [35]. Results from Mulroy et al. suggest that depression is related to a decrease in physical activity and wheelchair propulsion activities, while satisfaction with life is related to leisurely physical activities [34].
Compared with the individuals who transitioned from wheelchair use to walking or maintained wheelchair use or ambulation, the walking-towheelchair transition group had significantly lower QOL scores, including higher depression and higher pain severity [36]. Furthermore, one longitudinal study indicated that transitioning from ambulation to wheelchair use can negatively impact psychosocial health 10 years after SCI [37]. One recent meta-analysis study showed that higher self-efficacy had a strong negative correlation with fewer depressive (18 studies) and anxiety symptoms (7 studies), and showed a trend towards a positive correlation between self-efficacy and quality of life [38]. Therefore, self-efficacy seems an important target in the rehabilitation of patients living with SCI.
Compared to patients who had excellent recovery and had non-excellent recovery at 6 monthpost- trauma, there were statistically significant differences in JOA score at admission (p=0.012), BDI score within one month (p=0.001), and HRERS within one month (p=0.006), but no significant differences in age (p=0.072), body mass index (p=0.689), levels of SCI (p=0.687), ISS (p=0.060), and neurosurgical intervention (p=0.073) between the two groups (Table 3). Compared to the SF-36 scores between the two groups, the excellent recovery group had higher SF-36 than non-excellent recovery group from hospitalization to 6 month follow-up.
According our results, the BDI and SF-36 can be good physiological and psychological assessment tools to help SCI patients with expressing psychological feelings and adapting to physical changes. Also, the results of BDI and SF-36 can help medical staff caring for the SCI patients with providing higher levels of empathy or assistance. The MH domain and MCS were negatively correlated with a patient’s age, meaning that elder patients tend to have lower MH and MCS scores.
Limitations
The study had several limitations. The SF-36 used to assess QoL is a screening tool, and the results of this research will require systematic replications with specialized neuropsychological tests that assess those domains more comprehensively. A second limitation might be the use of BDI as the reference standard for the measurement of depressive symptoms in the present study. Nonetheless, the resulting measurements may not be comparable to those produced by the currently institutionalized instruments that ascertain the presence of a depressive disorder. The ‘gold’ standard measurement for a depressive disorder at the moment is the structured diagnostic psychiatric interview [39]. A further limitation was the lack of long term follow-up, which may lead to certain bias in our results. Longer follow-up and increased participant numbers may be required in future studies to confirm the usage of SF-36 and BDI in patients with acute cervical SCI.
Conclusions
Our findings suggested that all adults with SCI should receive a QoL and depression screen. It is expected that the results of this study can help clinicians to understand the relationship between depressive symptoms and QoL in patients with spinal cord injury. Furthermore, the screens may serve as a reference for follow-up care for the medical staff.
Acknowledgments
This study was supported by grants from Chang Gung Memorial Hospital (Research Projects CMRPG8D1391). None of the authors have any conflicts of interest associated with this study.
Funding sources
This study was supported by grants from Chang Gung Memorial Hospital (Research Projects CMRPG8D1391).
References
- Craig A, Tran Y, Wijesuriya N, et al. Fatigue and tiredness in people with spinal cord injury. J. Psychosom. Res 73(3), 205-210 (2012).
- Craig A, Perry KN, Guest R, et al. Adjustment following chronic spinal cord injury, Determining factors that contribute to social participation. Br. J. Health. Psychol 20(4), 807-823 (2015).
- Pollard C, Kennedy P. A longitudinal analysis of emotional impact, coping strategies and post-traumatic psychological growth following spinal cord injury, a 10-year review. Br. J. Health. Psychol 12(3), 347-362 (2007).
- Wijesuriya N, Tran Y, Middleton J, et al. Impact of fatigue on the health-related quality of life in persons with spinal cord injury. Arch. Phys. Med. Rehabil 93(2), 319-324 (2012).
- Post MW, van Leeuwen CM. Psychosocial issues in spinal cord injury, a review. Spinal. Cord 50(5), 382-389 (2012).
- Krause JS, Kemp B, Coker J. Depression after spinal cord injury, relation to gender, ethnicity, aging, and socioeconomic indicators. Arch. Phys. Med. Rehabil 81(8), 1099-1109 (2000).
- Chen HY, Boore JR. Establishing a super-link system, spinal cord injury rehabilitation nursing. J Adv Nurs 57(6), 639-648 (2007).
- Chen HY, Boore JR. Living with a spinal cord injury, a grounded theory approach. J. Clin. Nurs 17(5), 116-124 (2008).
- Kennedy P, Duff J, Evans M, et al. coping effectiveness training reduces depression and anxiety following traumatic spinal cord injuries. Br. J. Clin. Psychol 42(1), 41-52 (2003).
- Kennedy P, Rogers B, Speer S, et al. Spinal cord injuries and attempted suicide, a retrospective review. Spinal. Cord 37(12), 847-852 (1999).
- Lee Y, Mittelstaedt R. Impact of injury level and self-monitoring on free time boredom of people with spinal cord injury. Disabil. Rehabil 26(19), 1143-1149 (2004).
- Tam SF. Quality of life, theory and methodology in rehabilitation. Int. J. Rehabil. Res 21(4), 365-374 (1998).
- Clayton KS, Chubon RA. Factors associated with the quality of life of long-term spinal cord injured persons. Arch. Phys. Med. Rehabil 75(6), 633-638 (1994).
- Farquhar M. Elderly people's definitions of quality of life. Soc. Sci. Med 41(10), 1439-1446 (1995).
- Zhan L. Quality of life, conceptual and measurement issues. J. Adv. Nurs 17(7), 795-800 (1992).
- Ware JE, Sherbourne CD. The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection. Med. Care 30(6), 473-483 (1992).
- Kortte KB, Falk LD, Castillo RC, et al. The Hopkins Rehabilitation Engagement Rating Scale, development and psychometric properties. Arch. Phys. Med. Rehabil 88(7), 877-884 (2007).
- Beck AT, Brown GK, Steer RA. Psychometric characteristics of the Scale for Suicide Ideation with psychiatric outpatients. Behav. Res. Ther 35(11), 1039-1046 (1997).
- Benzel EC, Lancon J, Kesterson L, et al. Cervical laminectomy and dentate ligament section for cervical spondylotic myelopathy. J. Spinal. Disord 4(3), 286-295 (1991).
- Baker SP, O'Neill B, Haddon W, et al. The injury severity score, a method for describing patients with multiple injuries and evaluating emergency care. J. Trauma 14(3), 187-196 (1974).
- Hirabayashi K, Miyakawa J, Satomi K, et al. Operative results and postoperative progression of ossification among patients with ossification of cervical posterior longitudinal ligament. Spine 6(4), 354-364 (1981).
- Du W, Wang L, Shen Y, et al. Long-term impacts of different posterior operations on curvature, neurological recovery and axial symptoms for multilevel cervical degenerative myelopathy. Eur. Spine. J 22(7), 1594-1602 (2013).
- Vilagut G, Valderas JM, Ferrer M, et al. Interpretation of SF-36 and SF-12 questionnaires in Spain, physical and mental components. Med. Clin 130(19), 726-735 (2008).
- Onishi T, Nishikawa K, Hasegawa Y, et al. Assessment of health-related quality of life after radiofrequency ablation or laparoscopic surgery for small renal cell carcinoma, a prospective study with medical outcomes Study 36-Item Health Survey (SF-36). Jpn. J. Clin. Oncol 37(10), 750-754 (2007).
- Arslantas D, Ayranci U, Unsal A, et al. Prevalence of hypertension among individuals aged 50 years and over and its impact on health related quality of life in a semi-rural area of western Turkey. Chin. Med. J (Engl) 121(16), 1524-1531 (2008).
- Williams R, Murray A. Prevalence of depression after spinal cord injury, a meta-analysis. Arch. Phys. Med. Rehabil 96(1), 133-140 (2015).
- Craig A, Perry KN, Guest R, et al. Prospective study of the occurrence of psychological disorders and comorbidities after spinal cord injury. Arch. Phys. Med. Rehabil 96(8), 1426-1434 (2015).
- Craig A, Tran Y, Middleton J. Psychological morbidity and spinal cord injury, a systematic review. Spinal. Cord 47(2), 108-114 (2009).
- Kennedy P, Rogers BA. Anxiety and depression after spinal cord injury, a longitudinal analysis. Arch. Phys. Med. Rehabil 81(7), 932-937 (2000).
- Khazaeipour Z, Taheri-Otaghsara SM, Naghdi M. Depression Following Spinal Cord Injury, Its Relationship to Demographic and Socioeconomic Indicators. Top. Spinal. Cord. Int. Rehabil 21(2), 149-155 (2015).
- Kraft R, Dorstyn D. Psychosocial correlates of depression following spinal injury, A systematic review. J. Spinal. Cord. Med 38(5), 571-583 (2015).
- Hassanpour K, Hotz-Boendermaker S, Dokladal P, et al. Low depressive symptoms in acute spinal cord injury compared to other neurological disorders. J. Neurol 259(6), 1142-1150 (2012).
- Tate DG, Forchheimer MB, Karana-Zebari D, et al. Depression and pain among inpatients with spinal cord injury and spinal cord disease, differences in symptoms and neurological function. Disabil. Rehabil 35(14), 1204-1212 (2013).
- Mulroy SJ, Hatchett PE, Eberly VJ, et al. Objective and Self-Reported Physical Activity Measures and Their Association With Depression and Satisfaction With Life in Persons With Spinal Cord Injury. Arch. Phys. Med. Rehabil 97(10), 1714-1720 (2016).
- Ataoglu E, Tiftik T, Kara M, et al. Effects of chronic pain on quality of life and depression in patients with spinal cord injury. Spinal. Cord 51(1), 23-26 (2013).
- Riggins MS, Kankipati P, Oyster ML, et al. The relationship between quality of life and change in mobility 1 year postinjury in individuals with spinal cord injury. Arch. Phys. Med. Rehabil 92(7), 1027-1033 (2011).
- Hiremath SV, Hogaboom NS, Roscher MR, et al. Longitudinal Prediction of Quality-of-Life Scores and Locomotion in Individuals with Traumatic Spinal Cord Injury. Arch. Phys. Med. Rehabil 98(12), 2385-2392 (2017).
- Van Diemen T, Crul T, van Nes I, et al. Associations Between Self-Efficacy and Secondary Health Conditions in People Living With Spinal Cord Injury, A Systematic Review and Meta-Analysis. Arch. Phys. Med. Rehabil 98(12), 2566-2577 (2017).
- Sheehan DV, Lecrubier Y, Sheehan KH, et al. The Mini-International Neuropsychiatric Interview (M.I.N.I.), the development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J. Clin. Psychiatry 59(20), 22-33 (1998).