Research Article - (2018) Volume 8, Issue 6
Fatal Familial Insomnia: Insight of the Most Common Genetic Prion Disease in China Based On the Analysis of 40 Patients
- *Corresponding Author:
- Xiao-Ping Dong
National Institute for Viral Disease Control and Prevention, Chinese Center for Disease, Control and Prevention, Chang-Bai Rd 155, Beijing 102206, China
Tel: +86-10-58900815
Fax: +86-10-58900815
Abstract
Fatal familial insomnia (FFI) is one of the subtypes of human genetic prion diseases and is the most frequently identified genetic prion disease in China. In this study, the epidemiological, clinical, laboratory and genetic features of 40 Chinese FFI patients were systematically analyzed. Despite of the distribution in 12 provinces, apparently more cases located in Henan and Guangdong provinces. The median onset age was 51 year-old (from 19-70 y) and the gender ratio was 1:1.5 (M:F). 45% cases displayed clinical symptoms less than 50 y and 32.5% cases did not have definitely disease family history. Sleeping disturbances, mental problems and recognition disorders were most common foremost symptoms, in which sleeping disturbances and recognition disorders were significantly frequent in the patients with positive family history and those with the onset age ≥ 50 y, respectively. Sympathetic symptoms, such as excessive sweating, salivation and minor evening pyrexia, were commonly noticed. 85% patients showed hypertension and 57.5% had obvious weight loss. 37.9% cases were cerebrospinal fluid (CSF) 14-3-3 positive and 25% were CSF RT-QuIC positive. Sporadic Creutzfeldt-Jakob disease (sCJD) associated abnormalities on MRI were recorded only in 6 patients with the onset-age ≥ 50 y. 126 individual from 14 families conducted prion gene (PRNP) sequencing and 36 asymptomatic carriers of D178N mutation and M129M homozygous were identified, among them 7 mutation carriers from 5 families were in the parent-generation and 5 from 3 families were elder siblings of the probands. The median survival was 10 months varying from 5 to 30 months. This is the largest study on FFI patients in China, as well as in Asia, which is helpful to enrich the knowledge of FFI.
Keywords
Fatal familial insomnia, Genetic prion disease, PRNP sequencing, D178N mutation
Introduction
Fatal familial insomnia (FFI) is a special subtype of human genetic prion disease which prominent pathology is occurs mainly in the thalamus, showing severe astrogliosis and loss of neurons. Genetically, FFI is linked to a GAC to AAC point mutation (substitution from aspartic acid to asparagines) at codon 178 of the prion protein gene (PRNP) (D178N) [1,2]. Same as other human genetic prion diseases, including genetic or familial Creutzfeldt-Jakob disease (gCJD or fCJD) and Gerstmann-Sträussler- Scheinker diseases (GSS), FFI is an autosomal dominant heredopathy. However, clinically FFI displays completely different phenotype, characterized by a disordered sleep-wake cycle, dysautonomia and motor signs. Pathologically, instead of spongiform degeneration and PrPSc deposits that are frequently observable in the brains of sporadic CJD (sCJD), which is the predominant type of human prion disease, and majority of human genetic prion disease, FFI is characterized by severe neuron loss and active astrogliosis in some brain regions, especially thalamus [3,4].
FFI was firstly described in an Italian family in 1986 [5]. Afterwards, FFI cases have been reported in many countries and regions, as well as in different races. FFI may occur without family history, or with definite clinical family history and/or genetic evidence [6-9]. Based on the surveillance data for human prion disease in China, FFI is most frequently detected genetic prion disease [10,11]. Up to June 2017, 40 FFI cases were diagnosed by the center of CJD surveillance system under the framework of China CDC, making up about 33% of all diagnosed genetic prion diseases. In this study, the epidemiological, clinical, genetic and laboratory characteristics of these 40 FFI cases were comparatively investigated based on the disease family history, gender and the onsetage. The RT-QuIC reactive profiles of the CSF samples from 39 patients and those of the brain samples from 3 patients were also proposed.
Materials and Methods
▪ Study population and data collection
40 diagnosed D178N/M129M FFI cases via China CJD surveillance program were enrolled into this study. The general information, the clinical data, the results of MRI and EEG, and the results of CSF 14-3-3 and PRNP sequencing were obtained from the database of national CJD surveillance program. According to the protocols of the national CJD surveillance, all referred cases were conducted with PRNP sequencing. The geography distributions of the patients were determined on the places they registered as permanent addresses. EEG abnormality was recorded only with the presences of periodic sharp wave complexes (PSWC). MRI abnormality was recorded with the presences of high signal in caudate/putamen and/or symmetrical or dissymmetrical cortical ribbon syndrome on diffusion-weighted imaging (DWI). The survival time was evaluated from disease onset to death.
▪ Laboratory tests
The specimens of blood, CSF and postmortem brain tissues of the suspected CJD patients were collected by physicians or pathologists in the local hospitals. CSF samples were obtained by routine lumbar puncture and employed into Western blot for protein 14-3-3. The peripheral blood leukocytes were isolated and the whole DNAs were extracted for sequencing analysis of PRNP and polymorphism of codon 129, using an automatic genetic analyzer (ABI3130XL). To avoid of DNA contamination in PCR and misreading in sequencing, all identified mutations were repeated at least one time using new blood samples. The standard operation procedure (SOP) of each assay was documented in the national CJD surveillance program described previously [10,12].
▪ RT-QuIC
The detailed operating protocol was described elsewhere [13]. Briefly, 10 μl CSF sample was subjected into RT-QuIC reaction containing 10 μg purified recombinant hamster PrP protein (aa23-230), 10 mM PBS, 170 mM NaCl, 10 μM EDTA, 1 mM Thioflavin T (ThT), 0.001% SDS in final concentration, with a final volume of 100 μl. RT-QuIC was performed on FLUOStar Omega (BMG). The working conditions were temperature: 50ºC; vibration speed: 700 rpm; vibration/incubation time: 90/30 sec; total reaction time: 90 h. 10-8 diluted brain homogenate of the hamsters infected with scrapie strain 263K were used as positive control and the same diluted brain homogenate of normal hamster was used as negative control. The cut-off value was set as the average value of the negative controls plus 3 times of SD. Each sample was duplicated in parallel. The sample was considered as positive when at least two parallel wells revealed positive reactive curves.
▪ Statistical assays
The statistical analyses were performed using the SPSS 11.5 statistical software program. Categorical variables were compared using the Chi-Square test. Probabilities (p value) less than 0.05 were considered to be statistical difference and less than 0.01 were considered to be significant difference.
▪ Ethics Statement
Usage of the stored patients’ information and samples in China CJD Surveillance Center has been approved by the Research Ethics Committee of National Institute for Viral Disease Control and Prevention, China CDC. The written informed content of each case had been asked and signed by the family member or the relative of the patient according to the requirement of CJD surveillance.
Results
▪ General information
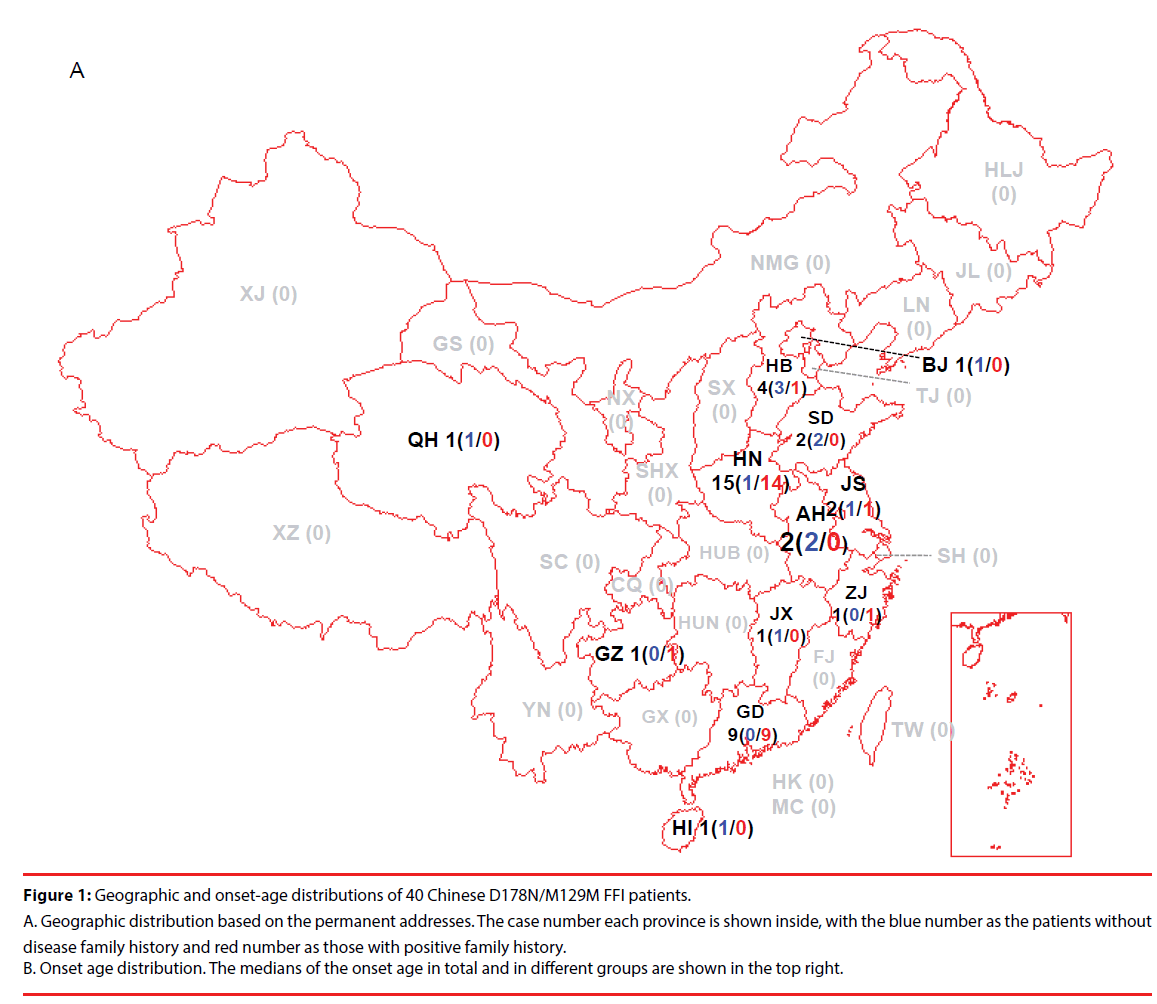
Up to June 2017, 40 FFI cases were recognized and diagnosed in China CJD Surveillance Center. All 40 FFI cases were Han-Chinese. According to the data of China CJD surveillance started officially since 2006, D178N FFI was the most commonly identified human gPrD in Chinese [10,11], making about 33% of all diagnosed gPrD. All 40 FFI were M129M homozygous. No polymorphisms of M129V or V129V were identified. Analysis of the permanent addresses of the patients revealed regional-associated tendency. 15 cases were from Henan (HN) province, 9 from Guangdong (GD), 4 from Hebei (HB), 2 from Shandong (SD), Jiangsu (JS) and Anhui (AH), 1 from Beijing (BJ), Zhejiang (ZJ), Jiangxi (JX), Guizhou (GZ), Qinghai (QH) and Hainan (HI), respectively (Figure 1A). The gender ratio (M: F) was 1:1.5 (16:24 cases). The onset ages varied from 19 to 70 year-old (y), with the median of 51 y. Majority of the patients (14 cases) was in the group of 50-59 y, followed by the groups of 40-49 y (7 cases) and 20-29 y (7 cases) (Figure 1B).
Figure 1: Geographic and onset-age distributions of 40 Chinese D178N/M129M FFI patients.
A. Geographic distribution based on the permanent addresses. The case number each province is shown inside, with the blue number as the patients without disease family history and red number as those with positive family history. B. Onset age distribution. The medians of the onset age in total and in different groups are shown in the top right.
27 out of 40 FFI cases (67.5%) had positive family histories obtained by careful interview and/or PRNP sequencing, while 13 (32.5%) cases did not (Table 1). Male patients distributed equally in the groups with and without family history (8:8 cases), while more female patients were in the group with family history (19:5 cases) but without statistical difference (P=0.0536). The onset ages of the cases without family history (median 42 y, ranging from 24-57 y) seemed to be younger than those with family history (median 53 y, ranging from 19-70 y). No FFI case without family history over 60 y was found (Figure 1B). Analysis of the onset ages between the groups with and without family history and between that of gender did not revealed statistical difference.
▪ Clinical features
The foremost symptoms of FFI cases varied largely, most patients appeared more than one symptom at onset. As shown in Table 1, mental problems (such as anxiety, dystropy, irritability and emotional lability) were reported in 50% (20/40) cases. Higher ratio of the cases without family history (61.5%, 8/13) described mental symptoms than those with family history (44.4%, 12/27). Sleep disturbances (such as insomnia and hypersomnia) were reported in 40% (16/40) cases, with more ratio in the cases with family history (51.6%, 13/27) than those without family history (15.4%, 2/13), showing statistical difference (P=0.0274). Also, 40% (16/40) of FFI recorded cognition disorders (such as memory decline, progressive dementia), showing 53.8% (7/13) in the cases without family history and 33.3% (9/27) in those with family history. 25% (10/40) cases described movement disturbances (such as gait instability), which distributing in 30.7% (4/13) cases without family history and 22.2% (6/27) cases with family history. Additionally, vision problems were reported in 4 cases, dizzy in 3 cases and fever in 3 cases. Cognition disorders were more frequently reported in the patients over 50 y (59.1%, 13/22) than those younger than 50 y (16.7%, 3/18), with statistical difference (P=0.0064). The positive ratios of mental problems, sleep disturbances, and movement disorders were 44.4%, 44.4% and 22.2% in the group of younger patients (<50 y), as well as 54.5%, 36.4% and 27.3% in that of older ones ( ≥ 50 y), respectively. No statistical differences in foremost symptoms were addressed between male and female cases.
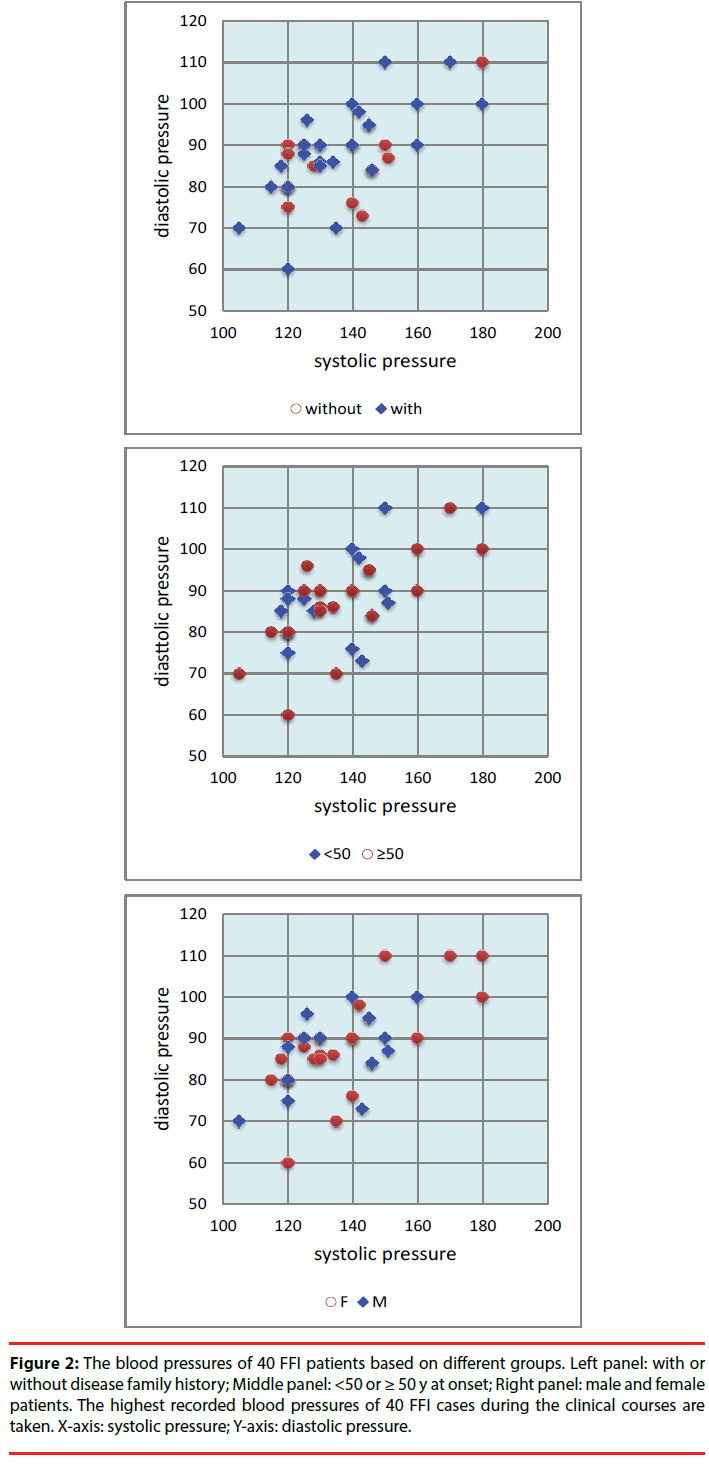
Along with the disease progression, more neurological symptoms and signs appeared. sCJD-associated symptoms (dementia, pyramidal and extrapyramidal symptoms, myoclonus, visual or cerebellar disturbance and mutism) were noticed in most of the patients during the clinical courses. Sympathetic symptoms were also reported frequently. Excessive sweating and minor evening pyrexia were observed in 55% (22/40) and 47.5% (19/40) patients, respectively. Eight cases (20%) recorded salivation clearly. Comparison of the sympathetic symptoms in the groups between the patients with and without family history, between the cases <50 and ≥ 50 y at onset, and between male and female patients did not reveal significant different (Table 1). Obvious weight loss was reported in 57.5% (23/40) cases, without statistical difference between various groups (Table 1). Hypertension was frequently noticed. Based on the normal values of blood pressure for various ages of Chinese, 85.0% FFI cases (34/40) showed high blood pressure (systolic and/or diastolic pressure) during the clinical courses. The younger patients looked to have higher ratio (94.4%, 17/18) of high blood pressure than the old ones (77.3%, 17/27), but without statistical difference (Table 1, Figure 2).
| Clinical manifestations | Total (n=40) |
Family history | Age of onset | Gender | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| With (n=27) |
Without (n=13) |
P value | <50 (n=18) |
≥50 (n=22) |
P value | Male (n=16) |
Female (n=24) |
P value | ||
| Age of onset (median) | 51 y (19-70) | 53 y (19-70) | 42 y (24-57) | 0.3403 | - | - | - | 50 y (24-62) | 51.5 y (19-70) | 0.6410 |
| Foremost symptoms | ||||||||||
| Mental problems | 20 (50.0%) | 12 (44.4%) | 8 (61.5%) | 0.3111 | 8 (44.4%) | 12 (54.5%) | 0.5250 | 9 (56.3%) | 11 (45.8%) | 05186 |
| Sleeping disturbances | 16 (40.0%) | 14 (51.6%) | 2 (15.4%) | 0.0274 | 8 (44.4%) | 8 (36.4%) | 0.8084 | 7 (43.8%) | 9 (37.5%) | 0.6926 |
| Recognition disorders | 16 (40.0%) | 9 (33.3%) | 7 (53.8%) | 0.2148 | 3 (16.7%) | 13 (59.1%) | 0.0064 | 6 (37.5%) | 10 (41.7%) | 0.7921 |
| Movement symptoms | 10 (25.0%) | 6 (22.2%) | 4 (30.7%) | 0.5587 | 4 (22.2%) | 6 (27.3%) | 0.7136 | 3 (18.8%) | 7 (29.2%) | 0.4560 |
| Sympathetic symptoms | ||||||||||
| Excessive sweating | 22 (55.0%) | 14 (51.9%) | 8 (61.5%) | 0.5640 | 11 (61.1%) | 11 (50.0%) | 0.4822 | 9 (56.3%) | 13 (54.2%) | 0.8967 |
| Salivation | 8 (20.0%) | 5 (18.5%) | 3 (23.1%) | 0.7356 | 4 (22.2%) | 4 (18.2%) | 0.7506 | 3 (18.8%) | 5 (20.8%) | 0.8717 |
| Minor evening pyrexia | 19 (47.5%) | 11 (40.7%) | 8 (61.5%) | 0.2173 | 10 (55.6%) | 9 (40.9%) | 0.3560 | 8 (50.0%) | 11 (45.8%) | 0.7960 |
| Weight loss | 23 (57.5%) | 17 (63.0%) | 6 (46.2%) | 0.9305 | 11 (61.1%) | 12 (54.5%) | 0.6760 | 10 (62.5%) | 13 (54.2%) | 0.6014 |
| High blood pressure | 34 (85.0%) | 23 (85.2%) | 11 (84.6%) | 0.9622 | 17 (94.4%) | 17 (77.3%) | 0.1302 | 14 (87.5%) | 20 (83.3%) | 0.7176 |
| CSF 14-3-3 | 14 (35.9%)a | 9 (34.6%)a | 5 (38.5%) | 0.8134 | 7 (41.2%)a | 7 (31.8%) | 0.5457 | 8 (50.0%) | 6 (26.1%)a | 0.1256 |
| CSF RT-QuIC | 10 (25.0%) | 7 (25.9%) | 3 (23.1%) | 0.8454 | 7 (38.9%) | 3 (13.6%) | 0.0665 | 5 (31.3%) | 5 (20.8%) | 0.4560 |
Table 1: The main clinical and laboratory features of Chinese FFI patients.
Figure 2: The blood pressures of 40 FFI patients based on different groups. Left panel: with or without disease family history; Middle panel: <50 or ≥ 50 y at onset; Right panel: male and female patients. The highest recorded blood pressures of 40 FFI cases during the clinical courses are taken. X-axis: systolic pressure; Y-axis: diastolic pressure.
▪ Clinical examination and laboratory tests
39 patients received EEG examination at least one time during hospitalization. None of them revealed typical PSWC. All 40 cases took MRI scanning and five of them showed the sCJDassociated abnormalities. Two cases showed high signal in caudate/putamen, one with high signal in right thalamus, one with high signals in caudate/putamen and bilateral thalamus, one with dissymmetrical cortical ribbon syndrome on DWI. Notably, all five cases with MRI abnormality were in the group of older patient (two 57 y, two 61 and one 65 at onset), showing statistical difference (P=0.0305). Western blot for CSF 14-3-3 was performed in 39 patients and the positive bands were detected in 35.9% (14/39) tested samples. Male patients in this study seemed to have higher positive rates (50%, 8/16) than the female (26.1%, 6/23), but without statistical significance (P=0.1256, (Table 1)).
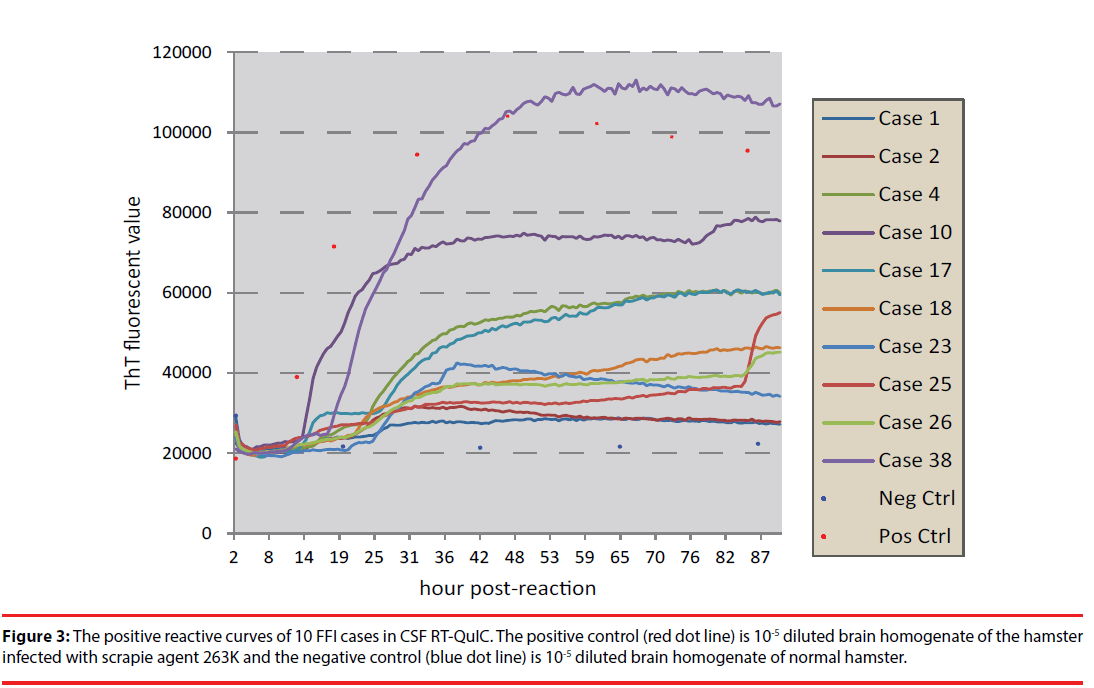
The CSF samples of 40 FFI patients were subjected into RT-QuIC assays using full-length recombinant hamster PrP (23-231) as substrate. Each sample underwent three times of RTQuIC. The patient showing positive reactions at least in two independent tests was considered to be positive in CSF RT-QuIC. Out of 40 cases, 10 (25%) were positive in CSF RT-QuIC. The converting times of the positive samples varied from 18 to 39 h post-reaction and the maximal ThT values were from 29500 to 110000 rfu (Figure 3). Analysis of the distribution of the cases with positive CSF RT-QuIC in the patients with and without family history, <50 and ≥ 50 y, and male and female did not Figure out any statistical difference (Table 1).
▪ Survival time
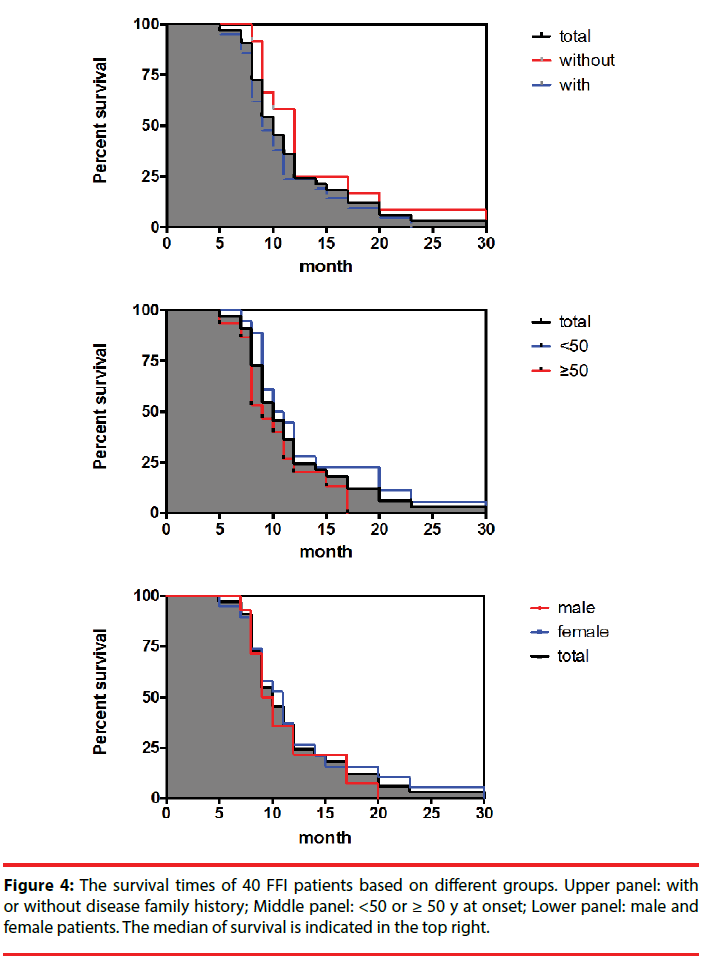
All FFI patients were carefully followed-up. Up to the end of June 2017, 33 cases have died with definite date, 4 were still alive and 3 were lost contact. The clinical durations of 33 dead cases varied largely from 5 months to 30 months, with the median of 10 months. 75.6% of the cases (25/33) died within one year after onset, while only one case lived longer than two years. Although the patients with family history, the patients <50 y at onset and female patients had relatively long median of survival, statistical assays did not show significance (Figure 4).
▪ PRNP sequencing and family history
All 40 FFI cases were confirmed to have a D178N mutation in one PRNP allele genetically, while methionine homozygous genotype at codon 129 (M129M). Clinical interviews a part of cases came from the same blood ties, but was reported and diagnosed independently. As described previously, Case 1 and Case 2 were uncle and niece [14]. Case 14 was half-blood aunt of Case 7, who appeared symptoms 3 years later at the age of 61. Case 24 (onset age: 27 y) was the niece of Case 11 (onset age: 42), who had clinical symptoms 3 years late.
Besides of the probands, totally 126 family members across 3 to 5 generations from 14 families also donated the blood samples for PRNP sequencing. The blood samples of all tested healthy individuals from one family were usually collected in the health section and transferred to our laboratory for sequencing assays simultaneously. Therefore, the tested samples contained the individuals whose parents (usually were proband’s siblings or uncle/aunt) were wild-type PRNP genotype verified by subsequent sequencing assays. Totally, 36 individuals were confirmed to be D178N carriers with M129M homozygous. Further, the tested family members and the mutation carriers were divided into different generations according to the proband’s blood ties, including the generations of grandparent, parent (including the parent and parent’s sibling), proband (proband’s sibling), son (proband’s own children) and grandson (proband’s own grandchildren). Only two grandparents (Case 6 and Case 33) conducted PRNP sequencing with wild-type genotype. In parent-generation, 15 individual received the tests and 8 (53.3%) contained the D178N mutation. In proband-generation, 17 proband’s sibling (besides of proband) were tested and 8 (47.8%) were D178N positive. In son- and grandson-generations, 18 and 15 individuals were tested and 9 (50.0%) and 6 (40.0%) were confirmed to have the D178N mutation, respectively. The rest five D178N carriers were the cousins or nephews of probands [14,15].
Except for one mutation carrier (the elder brother of Case 7) who appeared typical clinical symptoms and died more recently (not included in this study), the rest of the mutation carriers maintained healthy till the last follow-up (varied according to each case). 7 individuals from 5 families were in the parent-generation, including the mothers of Case 2, Case 5, Case 6 and Case 24, the father and two uncles and one aunt of Case 33. Five elder siblings of the probands of 3 families were also asymptomatic mutation carriers, including one elder sister of Case 1, two sisters of Case 2 and two brothers of Case 33.
Discussion
In this study 40 Chinese patients with D178N FFI are systematically analyzed for the features of epidemiological, clinical, laboratory and examination, genetic and family history, as well as survival time. To our knowledge, it is largest study of Chinese FFI patients. Based on our surveillance data for human prion diseases, more than 120 cases of genetic prion diseases have been identified, covering 15 different subtypes of PRNP mutations. D178N FFI is the most frequently observed genetic prion disease among Chinese [11]. In general, the epidemiological, clinical, laboratory characteristics of Chinese FFI patients are similar as Caucasian patients [4,6,16], with relatively early onset age, obvious sympathetic symptoms and relatively long clinical duration compared with the features of sCJD. High blood pressure and weight loss are also frequently observed.
Like the FFI cases reported worldwide [4,6,17,18], none of 40 Chinese patients recorded PSWC on EEG and a few of them appeared sCJD-associated abnormalities on MRI. More recently, Grau-Rivera et al have screened 15 patients with CJD and 5 patients with fatal insomnia (FI) using a so-called “3-Tesla magnetic resonance imaging scanner” and proposed the possibility of quantitative MRI in FI cases [19]. It might be an interesting topic for further observation. About 1/3 FFI cases were CSF 14-3-3 positive and 25% were CSF RT-QuIC positive, whose positive ratios are remarkably lower than that of another two most frequent Chinese genetic prion diseases, T188K gCJD and E200K gCJD (unpublished data). Compared with the data of the patients with E200K- and V201I-gCJD, apparently lower positive rate of D178N FFI cases in CSF RT-QuIC are also described elsewhere [20]. No correlation has been addressed between CSF 14-3-3 positive and CSF RT-QuIC positive.
Three FFI cases have been confirmed to have relative weak but definite proteinase K (PK)- resistant PrPSc in the postmortem brain tissues [14,15,21]. The RT-QuIC assays with the brain homogenates from temporal lobes of those three cases at the same experimental condition also show positive, even in the high dilution (10-7 diluted brain homogenates) (data not shown). However, only two of these three patients show the positive reaction in CSF RT-QuIC. The correlation between presence of brain PrPSc and reactivity in CSF RT-QuIC in FFI patients needs further study with more paired brain/CSF samples.
These 40 FFI cases distribute in 12 provinces based on their permanent addresses. However, there are marked more cases in Henan and Guangdong provinces, highlighting a geographic-associated tendency. Interestingly, most (14 out 15) of the cases from Henan province and all 9 cases from Guangdong province recorded positive family history. Even in a county of Henan province, 6 FFI cases with family history have been identified. Our further investigation have traced definite blood ties for six patients that belong to three large family with different surnames, despite of onset in different places and reported from different hospitals at different times. Such geographic and family clusters may highlight the possibility that the genotype of D178N/ M129M in Han-Chinese may have common origins of the chromosome carrying this mutation. Such regional-associated high incidences of FFI are also observed in some European countries, such as Italy and Spain [22,23]. However, since there were several large waves of migrations from Northern to Southern over last two thousands year in China history, it is extremely hard to Figure out such possibility, especially the patients between Henan and Guangdong provinces that are two thousands kilometers apart.
Asymptomatic carriers with D178N mutation are commonly found among the FFI families [14, 24]. In this study, 33 mutation carriers have been identified out of 114 tested individuals from 14 families. Except one person who appeared symptoms and died more recently, the rest mutation carries do not report to have the typical clinical manifestations even for more than 10 years after PRNP sequencing. Particularly, many asymptomatic carriers are the parents or the individuals of the last generation of the probands, or elder brothers and sisters of the probands. D178N mutation is described to be extremely strong pathogenicity, being consistent with up to 100% penetrance, together with other three mutations including P102L, A117V and E200K based on large population control cohorts [25]. Although D178N mutation causes spontaneous disease in mouse models [26-28], many studies of multiple multigenerational families have revealed asymptomatic carriers [9,24]. It is obvious that besides of special D178N mutation, other unknown factor(s) may also influence the onset of the disease.
FFI is one of the genetic prion diseases that show high ratio of positive family history. Based on the EUROCJD collaborative surveillance project from 1993 to 2002, 88% of FFI patients have positive family history [6]. A Germen study covering 41 FFI patients illustrates 65% positive family history [29]. Coincidental with the previous studies on Caucasian population, the about 2/3 (67.5%) Chinese FFI cases show definite positive family history. In contrast, FFI is not frequently reported in Japan [30] and all 4 Japanese FFI cases enrolled in the study finished by Minikel and the colleagues do not have family history [25]. FFI is also less frequent in Korea that only one case has been reported [31,32]. Certainly, determination of disease-associated family history depends largely on the careful inspection and communication clinically and good follow-up survey. Whether there are some unknown reasons that may affect the family history among different countries and races is not clear. Lacking of positive family history may increase the difficulty in the clinical diagnosis for FFI. Although some other clinical examinations, such as polysomnography and PET, seem to be helpful for FFI diagnosis clinically [18,29,33], they are not routinely accessible in a number of hospitals. Therefore, PRNP sequencing is still indispensable tool for the diagnosis of FFI.
Acknowledgements
We appreciate all staffs in China CJD surveillance system for their great work. We also thank all referring patients and their family members for supplying the required information and donating specimens. This work was supported by Chinese National Natural Science Foundation Grants (81630062, 81572048), National Key R&D Program of China (2016YFC1202700, 2017YFC1200500) and SKLID Development Grant (2015SKLID503 and 2016SKLID603).
References
- Chen C, XP Dong. Epidemiological characteristics of human prion diseases. Infect. Dis. Poverty 5(1), 47 (2016).
- Prusiner SB. The prion diseases. Brain. Pathol 8(3), 499-513 (1998).
- F Llorens, JJ Zarranz, A Fischer, et al. Fatal Familial Insomnia: Clinical Aspects and Molecular Alterations. Curr. Neurol. Neurosci. Rep 17(4), 30 (2017).
- P Montagna, P Gambetti, P Cortelli, et al. Familial and sporadic fatal insomnia. Lancet. Neurol 2(3), 167-76 (2003).
- E Lugaresi, R Medori, P Montagna, et al. Fatal familial insomnia and dysautonomia with selective degeneration of thalamic nuclei. N. Engl. J. Med 315(16), 997-1003 (1986).
- GG Kovacs, M Puopolo, A Ladogana, et al. Genetic prion disease: the EUROCJD experience. Hum. Genet 118(2), 166-74 (2005).
- M Schmitz, K Dittmar, F Llorens, et al. Hereditary Human Prion Diseases: an Update. Mol. Neurobiol 54(6), 4138-4149 (2017).
- K Sasaki, K Doh‐ura, Y Wakisaka, et al. Fatal familial insomnia with an unusual prion protein deposition pattern: an autopsy report with an experimental transmission study. Neuropathol. Appl. Neurobiol 31(1), 80-87 (2005).
- SHI Qi, C Cao, GAO Chen, et al. Clinical and familial characteristics of ten chinese patients with fatal family insomnia. Biomed. Environ. Sci 25(4), 471-475 (2012).
- C Gao, Q Shi, C Tian, et al. The epidemiological, clinical, and laboratory features of sporadic Creutzfeldt-Jakob disease patients in China: surveillance data from 2006 to 2010. PLoS. One 6(8), e24231 (2011).
- Q Shi, W Zhou, C Chen, et al. The Features of Genetic Prion Diseases Based on Chinese Surveillance Program. PLoS. One 10(10), e0139552 (2015).
- Q Shi, W Zhou, C Chen, et al. Quality evaluation for the surveillance system of human prion diseases in China based on the data from 2010 to 2016. Prion 10(6), 484-491 (2016).
- Q Shi, K Xiao, C Chen, et al. Clinical and laboratory features of 14 young Chinese probable sCJD patients. Prion 11(2), 128-135 (2017).
- XH Shi, J Han, J Zhang, et al. Clinical, histopathological and genetic studies in a family with fatal familial insomnia. Infect. Genet. Evol 10(2), 292-297 (2010).
- WL Xie, Q Shi, SL Xia, et al. Comparison of the pathologic and pathogenic features in six different regions of postmortem brains of three patients with fatal familial insomnia. Int. J. Mol. Med 31(1), 81-90 (2013).
- S Capellari, R Strammiello, D Saverioni, et al. Genetic Creutzfeldt-Jakob disease and fatal familial insomnia: insights into phenotypic variability and disease pathogenesis. Acta. Neuropathol 121(1), 21-37 (2011).
- JJ Zarranz, A Digon, B Atares, et al. Phenotypic variability in familial prion diseases due to the D178N mutation. J. Neurol. Neurosurg. Psychiatry 2005 76(11),1491-1496.
- P Cortelli, D Perani, P Montagna, et al. Pre-symptomatic diagnosis in fatal familial insomnia: serial neurophysiological and 18FDG-PET studies. Brain 129(3), 668-675 (2006).
- O Grau-Rivera, A Calvo, N Bargalló, et al. Quantitative Magnetic Resonance Abnormalities in Creutzfeldt-Jakob Disease and Fatal Insomnia. J. Alzheimers. Dis 55(1), 431-443 (2017).
- M Cramm, M Schmitz, A Karch, et al. Characteristic CSF prion seeding efficiency in humans with prion diseases. Mol. Neurobiol 51(1), 396-405 (2015).
- C Tian, D Liu, W Xiang, et al. Analyses of the similarity and difference of global gene expression profiles in cortex regions of three neurodegenerative diseases: sporadic Creutzfeldt-Jakob disease (sCJD), fatal familial insomnia (FFI), and Alzheimer's disease (AD). Mol. Neurobiol 50(2), 473-481 (2014).
- A Padovani, M D'alessandro, P Parchi, et al. Fatal familial insomnia in a new Italian kindred. Neurology 51(5), 1491-1494 (1998).
- AB Rodríguez-Martínez, C Barreau, I Coupry, et al. Ancestral origins of the prion protein gene D178N mutation in the Basque Country. Hum. Genet 117(1), 61-69 (2005).
- M Synofzik, P Bauer, L Schöls, et al. Prion mutation D178N with highly variable disease onset and phenotype. J. Neurol. Neurosurg. Psychiatry 80(3), 345-346 (2009).
- EV Minikel, SM Vallabh, M Lek, et al. Quantifying prion disease penetrance using large population control cohorts. Sci. Transl. Med 8(322), 322ra9 (2016).
- S Dossena, L Imeri, M Mangieri, et al. Mutant prion protein expression causes motor and memory deficits and abnormal sleep patterns in a transgenic mouse model. Neuron 60(4), 598-609 (2008).
- WS Jackson, AW Borkowski, H Faas, et al. Spontaneous generation of prion infectivity in fatal familial insomnia knockin mice. Neuron 63(4), 438-450 (2009).
- I Bouybayoune, S Mantovani, F Del Gallo, et al. Transgenic fatal familial insomnia mice indicate prion infectivity-independent mechanisms of pathogenesis and phenotypic expression of disease. PLoS. Pathog 11(4), e1004796 (2015).
- A Krasnianski, PS Juan, C Ponto, et al. A proposal of new diagnostic pathway for fatal familial insomnia. J. Neurol. Neurosurg. Psychiatry 85(6), 654-659 (2014).
- I Nozaki, T Hamaguchi, N Sanjo, et al. Prospective 10-year surveillance of human prion diseases in Japan. Brain 133(10), 3043-3057 (2010).
- MJ Lee, J Shin, EJ Chung, et al. Midbrain hypometabolism in fatal familial insomnia: a case report and a statistical parametric mapping analysis of a korean family. Case. Rep. Neurol 6(3), 243-250 (2014).
- BH Jeong, YS Kim. Genetic studies in human prion diseases. J. Korean. Med. Sci 29(5), 623-632 (2014).
- S Capellari, R Strammiello, D Saverioni. Hereditary Creutzfeldt-Jakob disease and fatal familial insomnia. Clin. Lab. Med 23(1), 43-64 (2003).