Research Article - (2018) Volume 8, Issue 4
Effects of the Nicotine Acetylcholine Receptors A3 Rs6495309 and B3 Rs6474412 Polymorphisms on Smoking Behavior in Young Men
- Corresponding Authors:
- Pei-Lain Chen
Department of Medical Laboratory Science and Biotechnology, Central Taiwan University of Science and Technology, No 666, Buzih Road, Beitun District, Taichung 40601, Taiwan
Tel: +886 958876864
Fax: +886 4 22396761
Prof. Ching-Shan Huang
Department of Clinical Pathology, Cathay General Hospital, No 280, Sec 4, Ren Ai Road, Taipei 10630, Taiwan
Tel: +886918458176
Fax: + 886228084060
Abstract
Objective
Although some effects of genetic polymorphisms related to nicotine-dopamine metabolism on smoking status have been identified, no variants of the nicotine acetylcholine receptor (CHRN) gene associated with smoking have been reported for the Taiwanese population. Hence, the aim of this study was to investigate the effects of the CHRNA3 rs6495309 and CHRNB3 rs6474412 polymorphisms on smoking behavior in young Taiwanese men.
Methods
Five hundred young Taiwanese men aged 20-25 years completed a self-report questionnaire, including the Fagerstrom Test for Nicotine Dependence (FTND), the Physiological Cigarette Dependence Scale (PCDS) and the Cigarette Withdrawal Scale (CWS-21), and data on the CHRNA3 rs6495309 and CHRNB3 rs6474412 polymorphisms were collected. We evaluated the effect of different genotypes on smoking status, smoking intensity, FTND, PCDS and CWS-21 in the study cohort comprised of young men.
Results
In a comparison of 243 never smokers and 230 current smokers, the odds ratios for the development of both smoking and heavier smoking were not statistically significant in all genotypes. The FTND score was significantly different between subjects carrying CHRNB3 rs6474412 CC and those possessing CHRNB3 rs6474412 TT [mean (SD): 6.20 (2.17) versus 3.57 (2.17), P=0.022], while the CWS-21 score of subjects with CHRNB3 rs6474412 TC was significantly different from that for CHRNB3 rs6474412 TT [57.01 (14.95) versus 49.70 (18.26), P=0.030]. The trend of an increased percentage of FTND scores ≧ 6 among the CHRNB3 rs6474412 TT subgroup, TC subgroup and CC subgroup was statistically significant (P<0.05).
Conclusion
This study demonstrates that FTND and CWS-21 scores are significantly different between the CHRNB3-rs6474412-variant group and the wild-type group. The CHRNB3 rs6474412 variant is a useful indicator for evaluating smoking behavior in young Taiwanese men.
Keywords
Cigarette withdrawal scale, Fagerstrom test for nicotine dependence, Nicotine acetylcholine receptor, Smoking behavior
Introduction
Tobacco smoking is a multi-factorial behavior with both genetic and environmental determinants [1]. While environmental influences play a strong role in the initiation of smoking and smoking persistence, smoking quantity and nicotine dependence are highly related to heritability [2,3], although psychiatric disorders may affect the Fagerstrom Test for Nicotine Dependence (FTND) scores of smokers [4]. Thus, the assessment of genetic background could be a promising tool for understanding smoking behavior and guiding the selection of the most effective cessation treatment for an individual smoker.
Nicotine is the major psychoactive ingredient in tobacco and combines with the acetylcholine receptor (CHRN) to modulate dopamine activity in the midbrain, contributing to the development and maintenance of rewarding behaviors such as smoking [3]. Smokers modulate their smoking to maintain brain nicotine levels within a certain concentration range, and factors that alter nicotine clearance affect smoking behavior [3]. Individuals who eliminate nicotine rapidly are less likely to achieve clearly low craving scores even after smoking freely [3]. Thus, genetic polymorphisms in both nicotine metabolism and dopamine catabolism genes may influence smoking behavior, interact with each other to modulate risk and affect smoking cessation therapies. It has been shown that genetic variants related to nicotine-metabolizing enzymes, the dopaminergic system and opioid receptors may play an important role in smoking behavior [5]. Thus, markers from every step of these pathways may be useful for personalizing the choice of smoking cessation treatment in smokers. Among these genes, cytochrome P450 2A6 (CYP2A6) and CHRN are the best studied in both adult and adolescent smokers [2,5,6].
Smoking addiction is currently a significant social problem in the Taiwanese population [7]. Prevention and cessation programs for smokers have been implemented at many hospitals in Taiwan. Three studies concerning the relationship between genetic polymorphisms and smoking behavior in the Taiwanese population have been reported: (1) For smokers within a group of methadone maintenance patients, polymorphisms of the μ-opioid receptor gene were found to be associated with the plasma concentration of cotinine [8], (2) The interaction of the dopamine D2 receptor TaqIB and monoamine oxidase A was found to affect smoking intensity in young men [9] and (3) A single nucleotide polymorphism (SNP) (rs4680) of the catechol-O-methyltransferase (COMT) gene and the interaction between the CYP2A6 and COMT genes were shown to affect smoking status in young men [10].
Within the Taiwanese population, a significant association between CHRNA7 rs8024987 polymorphisms (GG and GG+GC, respectively) and a better cholinesterase inhibitor response has been identified in patients with Alzheimer’s disease [11]. Additionally, a significant interaction was demonstrated between rs7179008 of the CHRNA7 gene (GT haplotype in block4) and late-onset Alzheimer’s disease risk [12]. However, no variants of the CHRN gene have been found to be associated with smoking behavior in the Taiwanese population.
In the CHRN gene, CHRNA3 rs6495309 and CHRNB3 rs6474412 are two common variants that have been studied in relation to smoking behavior and smoking-related diseases [13,14]. Therefore, in this pioneering study conducted in Taiwanese cohort, CHRNA3 rs6495309 and CHRNB3 rs6474412 were selected as candidate polymorphisms. Only men were invited to participate in this study because in women not using oral contraceptives, nicotine and cotinine clearance are 13% and 24% higher, respectively, than in men [6]. Furthermore, Taiwanese men smoke significantly more cigarettes than Taiwanese women [7,15].
Materials and Methods
▪ Subjects
Our cross-sectional study was advertised to all students at both Chang Jung Christian University, Tainan, Taiwan and Central Taiwan University of Science and Technology, Taichung, Taiwan. Volunteers contacted the authors of the study, and convenience samples were then screened for eligibility at the health centers of the two universities. All participants provided written, informed consent at the beginning of study, and the review board of Chang Jung Christian University (CJCU-99004) and Central Taiwan University of Science and Technology (CTUST-99016) approved this study. The study was conducted in accordance with good clinical practice procedures and the declaration of Helsinki.
A self-report questionnaire was given to all study subjects [16-18]. The questionnaire included demographic data, smoking background and status, and smoking-related surveys. We utilized the six-item FTND [16], the 15-item short form of the Physiological Cigarette Dependence Scale (PCDS) derived from the 30-item PCDS [18] and the Withdrawal Symptoms Scale [Cigarette Withdrawal Scale (CWS-21)] [17,18]. Biomarkers (nicotine and cotinine concentrations) are used for the validation of these questionnaires [16-18] with no overor under-exaggeration. Never smokers were defined as persons who had never smoked in their lifetime. Current smoking was defined as ever smoking cigarettes on one or more of the past 30 days. Ever smokers were defined as persons who smoked at one time, had quit, and were not currently smoking. Current smokers were divided into two groups: light smokers and heavy smokers, depending on smoking a number of cigarettes per day (CPD) lower than or equal to (or higher than), respectively, the average number among all smokers. The exclusion criteria were (1) a history of diagnosed mental health disease or cancer, (2) alcoholism or drug abuse, (3) severe communication problems, or (4) terminal illness. When the survey was completed, trained research assistants (licensed nurses or medical technologists) obtained blood samples at a university center.
▪ The determinations for CHRNA3 rs6495309 and CHRNB3 rs6474412 polymorphisms
For each study subject, five milliliters of whole blood with EDTA as an anticoagulant was required. Total genomic DNA was isolated from blood cells using a blood DNA isolation kit (Epicentre Corp., Taipei, Taiwan). The CHRNA3 rs6495309 and CHRNB3 rs6474412 polymorphisms were identified through polymerase chain reaction-restriction fragment length polymorphism (PCR-RFLP) analysis [19,20].
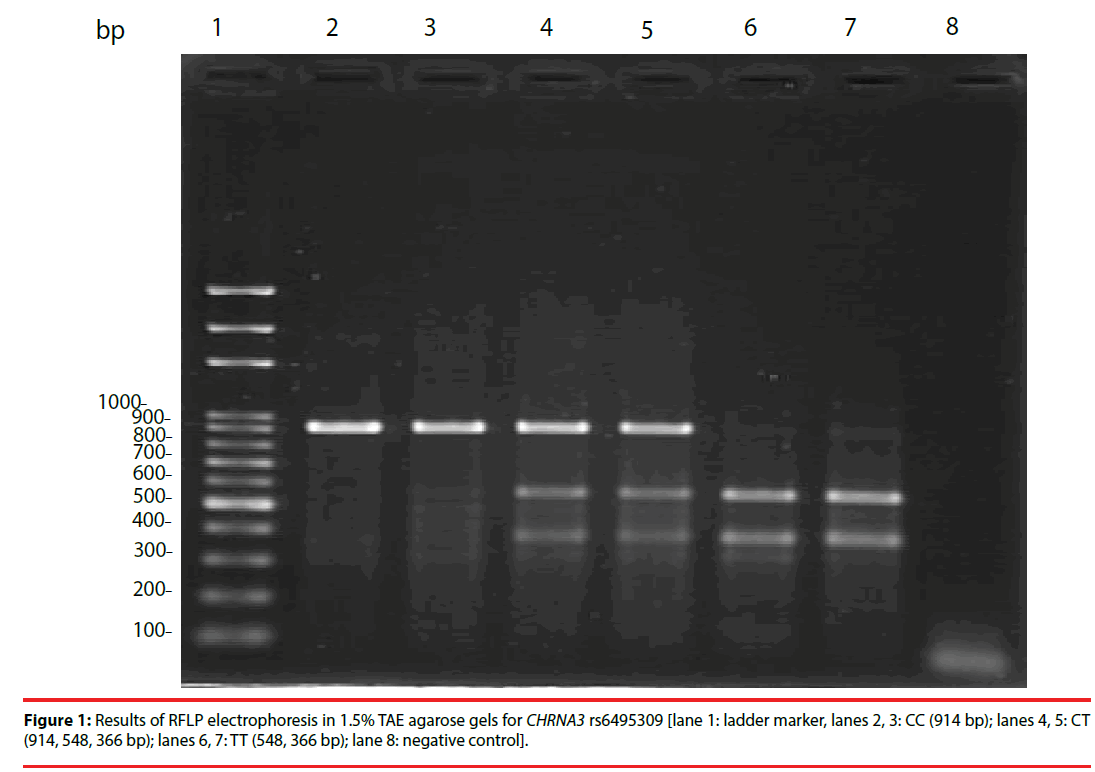
PCR was performed with a thermal cycler (Bio-Rad Corp., Carlsbad, CA, USA). The PCR conditions were as follows: first cycle, denaturation at 95°C for 5 min; cycles 2-35, denaturation at 94°C for 30 s, annealing at 59°C for 30 s, and elongation at 72°C for 30 s; and a final extension for 10 min at 72°C. For CHRNA3 rs6495309, we designed the forward primer 5’-CTC CTG GCA TTC AGC AA-3’ and the reverse primer 5’-AGG CGG CAG ATC ACC TAA-3’. We detected a 914-base pair (bp) fragment in 1.5% TAE agarose gels (Invitrogen Corp., Carlsbad, CA, USA) after electrophoresis at 100 V for 30 min. Using the restriction enzyme NlaIII, one fragment (914 bp) was observed for CC, three fragments (914, 548 and 366 bp) for CT and two fragments (548 and 366 bp) for TT. Positive controls including the CC, CT and TT genotypes of CHRNA3 rs6495309 were run for each of the genotyping assays.
For CHRNB3 rs6474412, the forward primer 5’-CCT CTT TTC CTG TGT CTA TTT GAT GGC-3’ and the reverse primer 5’-TTC ACC CTG CAA AGA TAC TCA TCA ACT CTT CAC C-3’ were used to amplify a 134- bp fragment, which was verified in 3.5% TAE agarose gels (Invitrogen Corp., Carlsbad, CA, USA) after electrophoresis at 100 V for 45 min. Using HaeIII as the restriction enzyme, one (134 bp), three (134, 109 and 26 bp) and two (109 and 26 bp) fragments were obtained for the TT, TC and CC genotypes, respectively. Positive controls including the TT, TC and CC genotypes of CHRNB3 rs6474412 were run for each of the genotyping assays.
▪ Statistical analysis
To assess the effects of genetic polymorphisms on smoking behaviors, CHRNA3 rs6495309 and CHRNB3 rs6474412 were analyzed separately. We evaluated the effect of different genotypes on smoking status, smoking intensity, FTND, PCDS and CWS-21 in the study cohort comprised of young men.
To evaluate the effect of the genetic variants on smoking status and smoking intensity, an odds ratio (OR) of 1 was assigned to subjects carrying the wild-type genotype. The Mantel–Haenszel Chi-square test was utilized to calculate the ORs and their 95% confidence intervals (CIs). To compare the significance of FTND, PCDS and CWS-21 among subjects carrying different polymorphisms of CHRNA3 rs6495309 and CHRNB3 rs6474412, the Kruskal-Wallis test was applied to compare quantitative data where appropriate. A P value of<0.05 was defined as statistically significant for each analysis. When the result of the Kruskal-Wallis test was statistically significant, Tukey multiple comparisons were performed. For trend analysis, the Cochrane- Armitage test was used. All data were analyzed using SPSS (version 18.0 software for Windows, SPSS Inc., Chicago, IL).
Results
▪ Distribution of genotypes
Five hundred men aged 20-25 years were recruited for this study. Analysis of the questionnaires revealed that there were 243 never smokers, 230 current smokers and 27 ever smokers in the cohort. Due to the small sample size, the 27 ever smokers were excluded from further study. The mean age did not differ significantly between the 230 current smokers and the 243 never smokers [mean (SD): 22.4 (1.72) years versus 22.2 (1.53) years, t=1.351, P=0.407, data not shown in the tables].
Examples of the PCR-RFLP results for CHRNA3 rs6495309 genotypes are shown in Figure 1, while those for CHRNB3 rs6474412 are similar to CHRNA3 rs6495309, excepted for smaller fragments (not shown in the figure). The CC, CT and TT genotypes of CHRNA3 rs6495309 and TT, TC and CC of CHRNB3 rs6474412 were all confirmed in each of the genotyping assays. As shown in Table 1, among the 243 never smokers, the distribution of both CHRNA3 rs6495309 (N=243) and CHRNB3 rs6474412 (N=240, missing data for three subjects) genotypes agreed with Hardy–Weinberg equilibrium (P>0.05, respectively, data not shown in the Tables).
| Smokers | Non-smokers | OR (95% CI)a |
P | |
|---|---|---|---|---|
| CHRNA3 rs6495309 CC | 62 | 61 | 1.0 | |
| CT | 126 | 127 | 0.98 (0.63~1.50) |
0.912 |
| TT | 42 | 55 | 0.75 (0.44~1.28) |
0.295 |
| CHRNB3 rs6474412 TT | 149 | 164 | 1.0 | |
| TC | 69 | 60 | 1.27 (0.84~1.91) |
0.261 |
| CC | 6 | 16 | 0.41 (0.16~1.08) |
0.072 |
aCalculated by the Mantel-Haenszel Chi-square test.
Table 1: Odds ratio for the effect of CHRNA3 rs6495309 and CHRNB3 rs6474412 on smoking status.
▪ Odds ratios for the development of smoking and heavier smoking
Table 1 also shows that, when never smokers were used as the reference group, the ORs of smoking for the subjects carrying variant genotypes were not statistically significantly greater than for the subjects possessing the wild-type genotype (P>0.05 for both CHRNA3 rs6495309 and CHRNB3 rs6474412, respectively).
The average number of CPD among the 230 current smokers was 10. Although data were missing for some subjects, when light smokers were employed as the reference group, the ORs for heavier smoking were not statistically significant for ether CHRNA3 rs6495309 or CHRNB3 rs6474412, as shown in Table 2 (P>0.05, respectively).
| Heavier smokers | Light smokers |
OR (95% CI)a |
P | |
|---|---|---|---|---|
| CHRNA3 rs6495309 CC | 28 | 33 | 1.0 | |
| CT | 52 | 70 | 0.88 (0.47~1.62) |
0.673 |
| TT | 18 | 24 | 0.88 (0.40~1.95) |
0.760 |
| CHRNB3 rs6474412 TT | 63 | 85 | 1.0 | |
| TC | 26 | 39 | 0.90 (0.50~1.63) | 0.726 |
| CC | 4 | 2 | 2.70 (0.48~15.20) | 0.260 |
aCalculated by the Mantel-Haenszel Chi-square test.
Table 2: Odds ratios for the effects of CHRNA3 rs6495309 and CHRNB3 rs6474412 on heavier smoking.
▪ Effects of gene polymorphisms on FTND, PCDS and CWS-21
The FTND, PCDS and CWS-21 scores of 154, 198, and 183 subjects, respectively, of the 230 current smokers were analyzed due to missing data. As shown in Table 3, among the three groups carrying TT, TC or CC of CHRNB3 rs6474412, the FTND and CWS-21 scores were both significantly different, respectively (P=0.041 and 0.009, respectively). The other four comparisons were not significantly different (P>0.05).
| FTND | PCDS | CWS-21 | ||||
|---|---|---|---|---|---|---|
| N | Mean (SD) | N | Mean (SD) | N | Mean (SD) | |
| CHRNA3 rs6495309 CC | 40 | 4.05 (2.11) | 50 | 36.08 (13.82) | 44 | 49.64 (14.13) |
| CT | 88 | 3.84 (2.19) | 114 | 36.58 (14.15) | 105 | 53.70 (18.98) |
| TT | 26 | 3.00 (2.02) | 34 | 34.88 (11.95) | 34 | 49.82 (16.56) |
| Pa | 0.111 | 0.879 | 0.393 | |||
| CHRNB3 rs6474412 TT | 97 | 3.57 (2.17) | 125 | 36.34 (13.74) | 118 | 49.70 (18.26) |
| TC | 47 | 3.85 (2.05) | 62 | 36.11 (13.05) | 55 | 57.02 (14.95) |
| CC | 5 | 6.20b (2.17) | 6 | 40.00 (22.29) | 5 | 52.20 (22.91) |
| Pa | 0.041c | 0.996 | 0.009d |
aCalculated by the Kruskal-Wallis test.
bFTND scores for the five subjects were 3, 6, 6, 7 and 9.
cTT versus CC, P=0.022 (by Tukey multiple comparisons).
dTT versus TC, P=0.030 (by Tukey multiple comparisons).
Table 3: Comparisons of FTND, PCDS and CWS-21 among smokers carrying different genotypes.
The results of Tukey multiple comparisons showed that the FTND score was significantly different between subjects carrying CHRNB3 rs6474412 CC and those possessing the TT (P=0.022), while the CWS-21 score was significantly different between subjects with CHRNB3 rs6474412 TC and those with TT (P=0.030). The percentages of subjects with an FTND score ≧ 6 in the CHRNB3 rs6474412 TT, TC and CC subgroups were 16.5% (16/97), 25.5% (12/47) and 80% (4/5), respectively and this trend of an increasing percentage of subjects with an FTND score ≧ 6 was statistically significant (P<0.05 by the Cochrane-Armitage test, data not shown in the Tables).
Discussion
CHRNA3 rs6495309 (C>T), located−2109 bp from the ATG transcription initiation site, is one of the most commonly studied SNPs of the CHRN gene, which maps to chromosome 15q25 [19]. The rs6495309 T and rs667282 T alleles of CHRNA5-A3-B4 are in high linkage disequilibrium (LD) (D’=0.90, r2 = 0.71) [19]. The rs6495309 functional SNP of the CHRNA3 gene affects the regulating of gene expression, leading to an increased lung cancer risk in Chinese populations [21,22]. This SNP is also associated with the risk of chronic obstructive pulmonary disease risk in Norwegian [23], Chinese [19,24] and Korean populations [25]. Furthermore, CHRNA3 rs6495309 has been shown to interact with smoking status and influence systolic blood pressure and body mass index in a Finnish population [26] and to affect survival in patients with early-stage non-smallcell lung cancer in a Korean population [27].
Very recently, the results of a comprehensive meta-analysis provided strong evidence of an association of CHRNA3 rs6495309 with lung cancer susceptibility [14]. The CHRNA3 rs6495309 C allele is thought to overlap with promoter histone marks and alter the regulatory motif [14]. In a functional study, the rs6495309 C allele was found to significantly increase CHRNA3 expression by altering the promoter binding ability of CHRNA3 to the transcriptional factor Oct-1 [21]. An investigation of whether SNPs can influence gene expression in cell lines expressing neuronal proteins or in small cell lung cancer cell lines, and whether nuclear proteins interact differentially with the alleles of each SNP revealed rs6495309 to be associated with nicotine dependence and the risk of lung cancer [28]. These studies indicated that CHRNA3 rs6495309 is an important SNP related to smoking behavior and smoking-related diseases. However, the results of our study showed that the variant status of CHRNA3 rs6495309 did not affect smoking status, smoking intensity, or FTND, or PCDS or CWS-21 score. We suspect that the reason for this lack of association is that the level of smoking is not sufficiently high in young Taiwanese men [the average number of CPD among current smokers is 10 (or average FTND score=3.75) 10, this manuscript], since different levels of exposure to smoking make no significant difference in the genotype and allele frequencies of CHRNA3 rs6495309 C>T [27].
Rs6474412 (T>C), located approximately 2.1 kb from the 5’ end of the CHRNB3 gene, is another commonly studied SNP of the CHRN gene, which maps to chromosome 8p1.1 [29]. The rs6474412 C, rs4593 G(T) and rs4952 T alleles are in high LD [2]. CHRNB3 rs6474412 has been associated with the number of CPD in smokers [2,13,30], FTND-dichotomized phenotypes [29], the Nicotine Dependence Syndrome Scale score [29], the Wisconsin Inventory of Smoking Dependence Motives score [29], heavy smoking [29], craving [29], lung cancer [2] and the severity of psoriasis vulgaris (in combination with smoking behavior) [31]. CHRNB3 rs6474412 can be tagged with variations elsewhere within the linked equilibrium block containing theα6 nAChR subunit gene (CHRNA6) [2,31]. Rs6474412 might increase the number of CPD via the CHRNB3 and CHRNA6 subunits [2,31]. The relationship between CHRNB3 rs6474412 and CPD appears to be closer than that between CHRNA3 rs6495309 and CPD.
It is assumed that nicotine receptor function is decreased in subjects carrying the homozygous rs6474412 CC variant compared with that in those possessing the wild-type genotype (TT), leading to lower levels of dopamine secreted in the brain when these two subject-groups smoke the same quantity of cigarettes. Therefore, more cigarettes might be required by subjects carrying the CC genotype than those with the wild-type genotype. It was, at least partially, for this reason that we observed that the FTND score differed significantly between the group homozygous for the CHRNB3-rs6474412 variant and the wildtype group in the present study.
Previous meta-analyses of adult smokers [aged 25-44 years, mean age (SD)=41.9 (13.3) years, respectively] of European, African American and Asian ancestry revealed that the variant rs6474412 allele is associated with smoking intensity (average CPD>20 cigarettes and average FTND score ≧ 6) [2,13,29,30]. Our study showed that such an association is detectable even in young men (aged 20−25 years) with an average CPD of 10 cigarettes (or average FTND score=3.75) (10, this manuscript), although the carriers of rs6474412 CC is only 2.7% in youngmen smokers. We also found that the variant rs6474412 allele is related to the percentage of FTND scores ≧ 6 as well as to the CWS-21 score. Therefore, we conclude that the CHRNB3 rs6474412 variant is a useful indicator for evaluating smoking behavior.
This study has some limitations. First, its sample size is small, and only five subjects carrying the CHRNB3 rs6474412 variant are observed. It needs further study with a larger sample size. Second, nicotine receptor function is hypothesized being decreased in subjects carrying the homozygous rs6474412 CC variant when compared with that in those possessing the wild-type genotype (TT). Further studies to prove the hypothesis are warranted.
In summary, CHRNB3 rs6474412 is useful for studying FTND and CWS-21 in smokers. The results obtained from this study will contribute to the development of an approach for reducing smoking among Taiwanese university students through effective smoking prevention and cessation intervention programs. These programs can then be applied to perform clinical studies of smokers at hospitals.
Acknowledgements
The work was supported by grants from the National Science Council of Taiwan (grant NSC–100–2320–B–371–001); Changhua Christian Hospital, Taiwan (grant CCH– ICO10008) and Central Taiwan University of Science and Technology (grant CTU104–P–18, CTU105–P–20).
Competing Interests
All authors declare that they have no competing interests.
References
- Quaak M, van Schayck CP, Knaapen AM, et al. Genetic variation as a predictor of smoking cessation success. A promising preventive and intervention tool for chronic respiratory diseases? Eur. Respir. J 33(3), 468-480 (2009).
- Thorgeirsson TE, Gudbjartsson DF, Surakka I, et al. Sequence variants at CHRNB3-CHRNA6 and CYP2A6 affect smoking behavior. Nat. Genet 42(5), 448-453 (2010).
- Do E, Maes H. Narrative review of genes, environment, and cigarettes. Ann. Med 48(5), 337-351 (2016).
- Wei HT, Lin WC, Chen YS, et al. Efficacy of varenicline for smoking cessation between smokers with and without psychiatric disorders: a 12-week observational study. Neuropsychiatry (London) 7(4), 458-465 (2017).
- Ware JJ, Munafo MR. Genetics of smoking behaviour. Curr. Top. Behav. Neurosci 23, 19-36 (2015).
- Benowitz NL, Hukkanen J, Jacob P. Nicotine chemistry, metabolism, kinetics and biomarkers. Handb. Exp. Pharmacol (192), 29-60 (2009).
- Health Promotion Administration. Taiwan tobacco control: annual report (2016).
- Chen YT, Tsou HH, Kuo HW, et al. OPRM1 genetic polymorphisms are associated with the plasma nicotine metabolite cotinine concentration in methadone maintenance patients: a cross sectional study. J Hum. Genet 58(2), 84-90 (2013).
- Huang CL, Ou WC, Chen PL, et al. Effects of Interaction between Dopamine D2 Receptor and Monoamine Oxidase A Genes on Smoking Status in Young Men. Biol. Res. Nurs 17(4), 1-7 (2015).
- Ou WC, Huang YC, Huang CL, et al. Interaction between cytochrome P450 2A6 and Catechol-O-Methyltransferase genes and their association with smoking risk in young men. Behav. Brain. Funct 13(1), 8 (2017).
- Weng PH, Chen JH, Chen TF, et al. CHRNA7 polymorphisms and response to cholinesterase inhibitors in Alzheimer's disease. PLoSOne 8(12), e84059 (2013).
- Weng PH, Chen JH, Chen TF, et al. CHRNA7 polymorphisms and dementia risk: interactions with apolipoprotein epsilon4 and cigarette smoking. Sci. Rep 6, 27231 (2016).
- Hubacek JA, Pankova A, Stepankova L, et al. SNPs within CHRNA5-A3-B4 and CYP2A6/B6 are associated with smoking dependence but not with tobacco dependence treatment outcomes in the Czech population. Gene 606, 35-38 (2017).
- Wang J, Liu Q, Yuan S, et al. Genetic predisposition to lung cancer: comprehensive literature integration, meta-analysis, and multiple evidence assessment of candidate-gene association studies. Sci. Rep 7(1), 8371 (2017).
- Huang CL, Cheng CP, Huang HW. The development of a Chinese-language instrument to measure social smoking motives among male Taiwanese smokers. J. Transcult. Nurs 24(4), 371-377 (2013).
- Huang CL, Lin HH, Wang HH. The psychometric properties of the Chinese version of the Fagerstrom Test for Nicotine Dependence. Addict. Behav 31(12), 2324-2327 (2006).
- Lu CC, Lin HH, Chen CJ, et al. Huang: Psychometric testing of the Chinese version of the Dimensions of Tobacco Dependence Scale. J. Clin. Nurs 18(17), 2470-2477 (2009).
- Huang CL, Lin HH, Wang HH. Wang: Cigarette dependence questionnaire: development and psychometric testing with male smokers. J Adv. Nurs 66(10), 2341-2349 (2010).
- Yang L, Qiu F, Lu X, et al. Functional polymorphisms of CHRNA3 predict risks of chronic obstructive pulmonary disease and lung cancer in Chinese. PLoSOne 7(10), e46071 (2012).
- Hubacek JA, Lanska V, Adamkova V. Lack of an association between SNPs within the cholinergic receptor genes and smoking behavior in a Czech post-MONICA study. Genet. Mol. Biol 37(4), 625-630 (2014).
- Wu C, Hu Z, Yu D, et al. Genetic variants on chromosome 15q25 associated with lung cancer risk in Chinese populations. Cancer. Res 69(12), 5065-5072 (2009).
- Xiao M, Chen L, Wu X, et al. The association between the rs6495309 polymorphism in CHRNA3 gene and lung cancer risk in Chinese: a meta-analysis. Sci. Rep 4, 6372 (2014).
- Pillai SG, Ge D, Zhu G, et al. A genome-wide association study in chronic obstructive pulmonary disease (COPD): identification of two major susceptibility loci. PLoS. Genet 5(3), e1000421 (2009).
- Cui K, Ge X, Ma H. Four SNPs in the CHRNA3/5 alpha-neuronal nicotinic acetylcholine receptor subunit locus are associated with COPD risk based on meta-analyses. PLoSOne 9(7), e102324 (2014).
- Lee JY, Yoo SS, Kang HG, et al. A functional polymorphism in the CHRNA3 gene and risk of chronic obstructive pulmonary disease in a Korean population. J. Korean. Med. Sci 27(12), 1536-1540 (2012).
- Kaakinen M, Ducci F, Sillanpaa MJ, et al. Associations between variation in CHRNA5-CHRNA3-CHRNB4, body mass index and blood pressure in the Northern Finland Birth Cohort 1966. PLoSOne 7(9), e46557 (2012).
- Jin G, Bae EY, Yang E, et al. A functional polymorphism on chromosome 15q25 associated with survival of early stage non-small-cell lung cancer. J. Thorac. Oncol 7(5), 808-814 (2012).
- Flora AV, Zambrano CA, Gallego X, et al. Functional characterization of SNPs in CHRNA3/B4 intergenic region associated with drug behaviors. Brain. Res 1529, 1-15 (2013).
- Chen LS, Baker TB, Grucza R, et al. Dissection of the phenotypic and genotypic associations with nicotinic dependence. Nicotine. Tob. Res 14(4), 425-433 (2012).
- Cui WY, Wang S, Yang J, et al. Significant association of CHRNB3 variants with nicotine dependence in multiple ethnic populations. Mol. Psychiatry 18(11), 1149-1151 (2013).
- Zhu KJ, Quan C, Zhang C, et al. Combined effect between CHRNB3-CHRNA6 region gene variant (rs6474412) and smoking in psoriasis vulgaris severity. Gene 544(2), 123-127 (2014).