Research Article - Neuropsychiatry (2017) Volume 7, Issue 5
Brain Dopamine Transporter Availability is Associated with Response Time (RT) Variability in Adults with ADHD
- Corresponding Author:
- Chin-Bin Yeh, MD, PhD
Department of Psychiatry
Tri-Service General Hospital
National Defense Medical Center, Taipei, Taiwan
Tel: +886-87927220
Fax: +886-87927221
Abstract
Objectives:
This study evaluated dopamine transporter availability (DAT) in the response time (RT) variability of adults with attention-deficit/hyperactivity disorder (ADHD).
Methods:
Twenty-four adults with ADHD and 12 healthy control subjects completed the Connors’ Continuous Performance Test (CPT) and were then screened with the Adult Self-Report Scale (ASRS). The severity of ADHD was assessed with the Conners’ Adult ADHD Rating Scales (CAARS). The sub-scores on the CPT were selected as the variables of RT variability. Pearson’s correlation was utilized to investigate the association of DAT and RT variability in adults with ADHD.
Results:
There were significant associations between RT variability and DAT availability concentration. More RT variability, including Hit reaction time standard error, Variability of Standard Error, Hit Reaction Time Block Change, and Hit Standard Error Block Reaction Time change, as well as attention problems (omission errors) and cognitive inflexibility (perseveration) on the CPT, were present in adults with ADHD compared to healthy controls.
Conclusions:
Abnormal brain dopamine transmission might be associated with greater RT variability, which might lead adults with ADHD to have inconsistent and poorer performance on higher-order cognitive activities.
Keywords
Dopamine, ADHD, DAT, Response time, Adult
Introduction
Research is gradually untangling the mystery of the neuropsychological bases of attention-deficit/ hyperactivity disorder (ADHD). Individuals diagnosed with ADHD have been described as consistently inconsistent [1], both in behavior and in performing neurocognitive tasks [2], and as a result face lifelong challenges in school, personal relationships, and in the work force as well.
One promising area for further study of ADHD is response time (RT) variability, which is often purported to indicate behavioral attention [3]. Individuals with ADHD show increased RT variability across a wide range of tasks, including tasks measuring reaction time on motor speed, choice decision, vigilance, behavioral inhibition, cognitive interference, working memory, and visual discrimination [4,5]. Children and adults with ADHD show significantly more variability in RT than do controls [6]. However, the mechanisms of RT variability in patients with ADHD are still unknown [6-8].
To better understand RT variability, researchers are beginning to examine its behavioral correlates. As Antonini et al. have noted, children with ADHD have slower mean RT and greater RT variability times than do control groups, and RT variability is not modulated by age, gender, or the presence of anxiety or depression [4].
Results of some studies have shown that executive deficits such as working memory, inhibition, and delayed aversion [9,10] are related to ADHD; however, other studies have failed to show a relationship between ADHD and delayed aversion [9,11]. RT variability was found to be strongly related to ADHD [12,13] and was not attributed to other executive deficits in patients with ADHD [13]. RT variability often leads patients with ADHD to have an irregular rhythm of response to tasks stimuli and potentially affects their performance.
Dopamine transporter (DAT) imaging is reliable for studying human adults’ brain pathology [14]. SPECT with [99m] Tc-TRODAT-1 as a ligand was used to evaluate striatal DAT in 14 healthy young men. Specific uptake ratios (SUR) of the striatum (ST) and its sub-regions, the caudate (CA) and the putamen (PU), were measured using the occipital cortex as the reference tissue. The reliability of the two measurements between test and retest showed significant correlations for the ST, CA, and PU. Thus, [99m] Tc-TRODAT-1 SPECT might provide a reproducible and reliable tool in clinical management of young patients with DATrelated disorders.
Dopamine transporter (DAT) imaging in patients with neuropsychiatric disorders is a promising technique that may help clarify RT variability. There is continuing support for the hypothesis that catecholamine dysregulation and associated fronto-striatal dysfunction underlie ADHD [15]. Researchers have suspected that the dopaminergic system is involved in RT variability because patients with ADHD can be successfully treated with methylphenidate (MPH), a potent DAT blocker [16]. Abnormal DAT is found to be related to many neuropsychiatric disorders, for example, ADHD with Tourette syndrome (TS) [17]. The deficits in DAT activity in patients with Tourette syndrome (TS) were observed using [99m]Tc-TRODAT-1. Functional abnormality of the dopamine system in patients with TS might be evident only in its early stages, and adaptation to tic symptoms might play a role in regulating the neural system. In our 2007 study, we investigated the complex dysregulation of the dopaminergic neurotransmitter system in 8 drug- naïve TS patients challenged with methylphenidate (MPH) [17]. DAT-binding ratios decreased significantly after MPH treatment in both patients and controls; however, a significant interaction between group and MPH effects was found only in the right caudate, which was mainly due to a smaller decline of the DAT-binding ratio after MPH administration in the TS group. The change in the DAT-binding ratio might indicate a functional abnormality of the dopaminergic system in the right caudate nucleus of TS patients.
Costa used DAT imaging to reflect brain inhibitory capacity [18] and impulsivity [19]. Results of Costa and colleagues’ studies [18,19] also suggested that neural MPH effects interact with the saliency of the behavior. Spencer, et al. [15] measured DAT binding with a highly selective radioactive ligand (11C-altropane) and PET scans in a group of 47 well-characterized, treatment-naïve, nonsmoking, non-comorbid adults with and without ADHD. The authors reported significantly increased DAT binding in the right caudate in the adults with ADHD compared with the matched controls. Theirs results confirmed abnormal DAT binding in the striatum of adults with ADHD, and provide further support that dysregulation of DAT may be an important component of the pathophysiology of ADHD.
Frontal-striatal brain circuitry has also been shown to be involved in RT variability [20]. The hypothesis that brain striatal dopamine transporter (DAT) is involved in the pathogenesis of ADHD is based on the efficacy of treatment with MPH, which reduces intra-individual RT variability in persons with ADHD [21]. In contrast, psychostimulants for ADHD, such as atomoxetine, or antidepressants like citalopram, whose main mechanisms do not focus on striatal dopamine, have not been shown to modulate RT variability [22].
To our knowledge, no previous study has investigated the relationship between DAT availability and RT variability among patients with ADHD. Therefore, we designed a study to investigate the role of DAT availability in RT variability among young adults with ADHD. We hypothesized that the results from the CPT could be used as dependent variables, and brain DAT, measured by SPECT, could be used as independent variables.s
Methods
▪ Participants
A group of 24 young adults with ADHD and 12 healthy young adult controls were recruited for the study from the outpatient population of a medical center. All subjects were recruited after giving informed consent, and a parent or guardian also signed informed consent for each ADHD participant. We considered the statistical power (24 ADHD patients vs. 12 healthy controls) sufficient to differentiate the phenotype (such as ADHD severity and impaired performance on neuropsychological tests) between individuals with ADHD and healthy controls. The healthy controls were interviewed with semi-structured questionnaires, to screen out neuropsychiatric disorders, medical disorders, and any history of substance use. Because there is a low prevalence of substance use in Asia compared to that in Western countries, urine testing was not used to screen for substance use among the healthy controls.
All subjects participated in a clinical interview that included the Chinese ADHD Adult Self- Report Scale, conducted by an experienced child psychiatrist. Patients who had a history of neurologic disease, schizophrenia, substance abuse or dependence, use of a psychotropic drug in the previous 6 months, or an estimated IQ lower than 70 were excluded from the study.
Screening for ADHD was done with the Adult Self-Report Scale (ASRS) for ADHD, and the severity of ADHD was assessed with the Conners’ Adult ADHD Rating Scales (CAARS). Participants were also assessed with neuropsychological tests, including the Conners’ Continuous Performance Test (CPT). They were studied with single photon emission computed tomography (SPECT) examinations using 99mTc-TRODAT-1 as a radioligand to measure the dopamine transporter (DAT) availability in the brain. This investigation was approved by the Ethics Committee at our hospital, the Tri- Service General Hospital, National Defense Medical Center, Taipei, Taiwan. All subjects were college students (19-22 years of age) or master’s level students (23-26 years of age).
▪ Measures
Chinese ADHD Adult Self-Report Scale: The ASRS is part of the World Health Organization’s Composite International Diagnostic Interview (CIDI), and we established its validity and reliability in a previous study [23]. The questionnaire consists of subscales for inattention and hyperactivity or impulsivity. Each subscale contains nine indicators that have persisted for at least 6 months ‘to a degree that is maladaptive and inconsistent with the patient’s developmental level.’
Conners’ Adult ADHD Rating Scales: The Conners’ Adult ADHD Rating Scale (CAARS) [24] is a quantitative measure of current ADHD symptomatology that can be completed by adult participants. It was designed to incorporate the manifestations of ADHD in adults based on the scientific literature and clinical experience.
Kiddie-Schedule for Affective Disorders and Schizophrenia, epidemiology version (K-SADS-E): A diagnosis of ADHD was based on clinical psychiatric interviews by the investigators using the Chinese version of the K-SADS-E. Previous studies have shown that the Chinese K-SADS-E is a reliable and valid instrument to assess child psychiatric disorders in Taiwan, and it has been used extensively in a variety of studies [25].
Conners’ Continuous Performance Test- Second Edition (CPT-II): In the CPT-II Test 24, respondents are asked to press a space bar on a computer keyboard when any letter other than the target letter “X” appears on a computer screen [26]. Stimuli are presented in six blocks with three sub-blocks, each containing 20 trials. The inter-stimuli stimulus intervals (ISI) are 1, 2, or 4 seconds, while the display time is held constant at 250 milliseconds (msec).
Several parameters were used in our study. The number of omission errors indicated the number of targets to which the subjects did not respond. The number of commission errors referred to the number of times that individuals responded to a non-target. Hit Reaction Time was the mean response time for all correct target hits during administration of the CPT-II. Perseveration was defined as a response that occurred within less than 100 msec following a stimulus, and indicated impulsivity, random response, and inattentiveness. Variability was a measure of how much an individual’s hit reaction time varied over the entire length of the test, thus providing a measure of intra-individual variability. Hit Reaction Time Block Change was the slope of change in reaction time over the six time blocks. A positive slope indicated a slowing reaction time, while a negative slope indicated quicker reaction times as the test progressed. Hit Standard Error Block Change was the slope of the change in reaction time standard error over the six time blocks. A positive slope indicated that reaction time became less consistent as the test progressed, while a negative slope indicated that reaction time became more consistent as the test continued. Both Hit Reaction Time Block Change and Hit Standard Error Block Change were considered indicators of loss of vigilance. Hit Reaction Time Inter-Stimuli Stimulus Interval Change was calculated by computing the slope of change in reaction time over the three Inter- Stimuli Stimulus Intervals (1, 2, and 4 seconds). It indicated a slowing of reaction time as the time between targets increased.
Hit Standard Error Inter-Stimuli-Stimulus Interval Change indicated erratic response as the time between targets increased. Response style (beta value) indicated avoidance of commission errors. Attentiveness indicated how well the individual discriminated between targets and non-targets. The variables were analyzed in keeping with the normative data for age and gender (initially provided in percentiles), and the results of this analysis were converted to percentiles (based on the test standardization sample) for subsequent analysis.
Measurement of brain dopamine transporter density using a radiopharmaceutical 99mTc TRODAT-1 on SPECT exam: A newly developed 99mTc-labeled tropane derivative, 99mTc TRODAT-1, [2-[[2-[[[3-(4-chlorophenyl)- 8-methyl-8-azabicyclo[3,2,1]oct-2-yl]methyl] (2- mercaptoethyl) minoethyl] amino] ethanethiolato (3-)- N2,N2′,S2,S2′]oxo-[1R- (exo-exo)], was modified by Kung, et al. [27] The derivative was prepared by reconstituting a preformulated lyophilized vial (provided by the Institute of Nuclear Energy Research, Lungtan, Taiwan) with 1110 megabecquerels (MBq) of freshly eluted 99mTc pertechnetate in 5 mL saline. Labeling was completed following heating of the reconstituted solution at 121° C for 30 minutes.s The 99mTc TRODAT-1 was obtained in a neutral solution (pH: 7.0–7.5) with >90% radiochemical purity over 6 hours, as determined by highperformance liquid chromatography. The shelf life of the lyophilized kit was >2 months at room temperature. 99mTc TRODAT-1 has been shown to selectively bind to DATs, and to be safe and useful in 1-day protocols for reconstructing SPECT images for daily routine application [28]. Available studies reported that 99mTc TRODAT-1 reliably assessed the DAT status of healthy rats, baboons, and humans using highcontrast SPECT 4 hr after injection [29].
Imaging acquisition and data analysis: For the imaging study, all participants were placed supine, with their heads fixed in a holder and aligned with an installed laser beam. To prevent any significant head movement during scanning, the subject’s head motion was also monitored by an external digital camera and an internal imaging quality check using sinogram and linogram. Brain SPECT images were acquired 4 hr after a single bolus injection of 740 MBq of 99mTc TRODAT-1 into the cephalic vein. A dual-head camera equipped with ultra-high-resolution fanbeam collimators (GE Millennium VG Hawkeye Dual Head Nuclear Camera, MillenniumTM VG, General Electric Corp, Milwaukee, WI, USA) was used. The acquisition time required to complete a brain SPECT study was about 30 minutes.
Data were acquired in a 128×128 matrix through 360° rotation (180° for each head) at 3° intervals for 30 seconds per angle step, resulting in a pixel size of 3.17×3.17 mm (in a 1.4 zoom) and a slide thickness of 3.4 mm. Images were reconstructed using back projection with a modified Metz filter (power: 3.5; cutoff frequency: 0.28 cycles/ pixel). Attenuation correction was performed using Chang’s first-order method (attenuation coefficient, μ=0.12 cm-1).
SPECT images were obtained at the level of the canthomeatal line. The resolution on the reconstructed SPECT images (mm full-width at half-maximum [FWHM] ) was 2 mm x 2 mm x 2 mm. Regions of interest (ROIs) in the right and left caudate and putamen were marked and guided by the corresponding magnetic resonance image (MRI). Regions were marked on composite images of the three sections, depicting the basal ganglia with the highest level of activity. The ROIs on one side were then transferred to the other side. The occipital cortices were drawn in the same way (e.g., the medial parts of bilateral occipital lobes at the basal ganglia level) and served as background areas. The averaged counts acquired per slice at the basal ganglia level were around 0.34 Mcounts. The numbers of pixels in the ROIs were approximately 57 for the caudate, 115 for the putamen, and 210 for the occipital cortex. The DAT-binding ratio was calculated by subtracting the mean counts per pixel in the occipital cortex from the mean counts per pixel in the whole striatum, putamen, or caudate nucleus, and then by dividing the result by the mean counts per pixel. The method is as follows: (target−occipital cortex) /occipital cortex, where target represents the striatum, putamen, or caudate nucleus [14].
Statistical analysis
Demographic data of age and gender were represented as mean ± standard deviation (SD) and n (%), respectively, for ADHD patients and healthy controls. Our method of analysis was reviewed and examined by biostatistician Dr. Meei-Shyuan Lee from the Department of Public Health, National Defense Medical Center. Differences between groups by sex and gender were compared using a two-sample t-test and Fisher’s exact test, respectively. Data for continuous performance test (CPT) examination, ADHD severity, and DAT availability were either summarized as (1) mean ± SD by group and then compared by using a two-sample t-test if the data followed normal distribution, or (2) summarized as median (IQR) by group and compared by using a Mann-Whitney U test if the data didn’t follow a normal distribution pattern. Pearson’s correlation analysis was carried out to assess the correlation between CPT examination and DAT availability, but Spearman’s correlation analysis was conducted if the data did not follow a normal distribution pattern. Results were shown as coefficient of correlation (r) with corresponding significance of the p-value. All statistical assessments were two-tailed and considered significant as p <0.05. All statistical analyses were carried out with IBM SPSS statistical software version 22 for Windows (IBM Corp, Armonk, NY, USA).
Results
A total of 24 young adults with ADHD (14 males, 10 females) and 12 healthy controls (10 males, 2 females) were recruited for the study. Both groups had similar distributions of age and gender (Table 1). Although higher dopamine transporter density in females than in males was reported in a previous study by Lavalaye, et al. [30], in our study there was no difference in gender distribution between the two groups (Table 1; p= 0.260). The average ages for ADHD patients and healthy controls were 27.1 yr (SD=4.5 yr), and 24.6 yr (SD=2.9 yr), respectively. All participants completed the study.
| Variables | ADHD patients (n=24) |
Healthy controls (n=12) |
P-value |
|---|---|---|---|
| Age (yr) | 27.1 ± 4.5 | 24.6 ± 2.9 | 0.093 |
| Gender | 0.260 | ||
| Females | 10 (41.7) | 2 (16.7) | |
| Males | 14 (58.3) | 10 (83.3) |
Age was represented as mean ± SD and compared using a two-sample t-test; gender was represented as n (%) and compared using Fisher’s exact test. No significant difference was noted between groups.
Table 1: Demographic data of ADHD patients and healthy controls.
Table 2 shows the comparisons of results from the CPT examination and the severity of symptoms among ADHD patients and healthy controls. The healthy controls performed significantly better on the CPT examination than did the ADHD patients. This was true for omission, hit reaction time (Hit RT), hit reaction time standard error (Hit RT SE), variability of standard error (variability of SE), perseveration, hit reaction time block change (Hit RT Block Change), and hit standard error reaction time block change (Hit SE RT Block Change) (all p<0.05). The degree of ADHD severity was significantly different between the two groups in total score, inattention, hyperactivity/impulsivity and ADHD index (all p<0.05).
| Variables | ADHD patients (n=24) |
Healthy controls (n=12) |
P-value |
|---|---|---|---|
| CPT examination | (n=21) | (n=12) | |
| Omission | 14.19 ± 25.10 | 35.90 ± 30.08 | 0.034* |
| Commission | 48.72 ± 28.69 | 56.50 ± 27.15 | 0.451 |
| Hit RT | 0 (0 , 4.09) | 10.24 (1.32 , 26.24) | 0.009* |
| Hit RT SE | 6.26 ± 12.36 | 23.71 ± 24.81 | 0.039* |
| Variability of SE | 5.88 ± 10.44 | 28.46 ± 29.47 | 0.024* |
| DPrime | 70.37 ± 23.98 | 60.24 ± 23.84 | 0.453 |
| Beta | 32.01 ± 10.64 | 36.18 ± 11.98 | 0.522 |
| Perseveration | 10.34 ± 18.27 | 41.43 ± 31.34 | 0.001* |
| Hit RT Block Change | 8.90 ± 18.20 | 41.63 ± 26.63 | <.001* |
| Hit SE Block Change | 17.38 ± 29.74 | 56.60 ± 33.49 | 0.002* |
| Hit RT ISI Change | 16.79 ± 28.82 | 32.80 ± 23.59 | 0.112 |
| Hit SE ISI Change | 13.33 ± 23.97 | 32.10 ± 31.42 | 0.063 |
| CAARS | (n=24) | (n=12) | |
| Total score | 63.83 ± 10.16 | 16.92 ± 13.66 | <.001* |
| Inattention | 22.17 ± 3.60 | 6.42 ± 4.46 | <.001* |
| Hyperactivity/Impulsivity | 17.38 ± 5.51 | 7.00 ± 5.75 | <.001* |
| ADHD index | 24.29 ± 4.12 | 3.50 ± 3.58 | <.001* |
Abbreviations: CPT, continuous performance test; ADHD, attention-deficit/hyperactivity
disorder; DAT, dopamine transporter availability; Hit RT, Hit reaction time; Hit RT SE, Hit reaction
time standard error; Variability of SE, variability of standard error; DPrime, measures how well the
individual discriminates between targets and nontargets; Beta, Response styles (this statistics
allows for evaluation of the speed/accuracy trade off); Hit RT Block Change, Hit Reaction Time
Block Change; Hit SE Block Change, Hit Standard Error Block Change; Hit RT ISI Change, Hit
Reaction Time ISI Change; Hit SE ISI Change, Hit Standard Error ISI Change; CAARS, Conners’
adult ADHD Rating Scales.
Data were represented as mean±SD and compared using two-sample t-test if data followed
normal distribution or median (IQR) and compared using Mann-Whitney U test if data didn’t
follow normal distribution.
*p<0.05 indicates significant difference between groups.
Table 2: Comparison of the results of CPT examination and ADHD severity between ADHD patients and healthy controls.
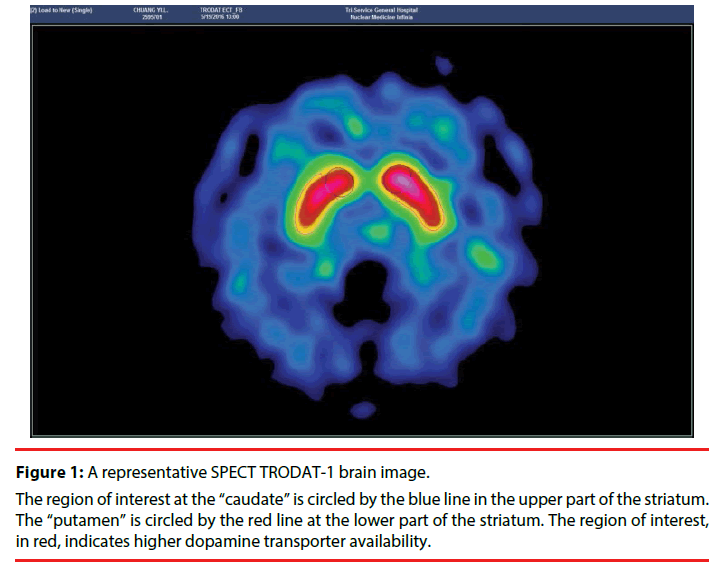
The comparison of 99mTc labeled tropane derivative, [2-[[2-[[[3-(4-chlorophenyl)- 8-methyl-8-azabicyclo[3,2,1]oct-2-yl]methyl] (2- mercaptoethyl) mino]ethyl]amino] ethanethiolato (3-)- N2,N2′,S2,S2′]oxo- [1R-(exo-exo)] (TRODAT) variables of the DAT availability between ADHD patients and healthy controls is shown in Table 3. DAT availability was similar between ADHD patients and healthy controls at all given time points (all p > 0.05). Figure 1 shows a representative SPEC TRODAT brain image with dopamine transport availability.
| [99mTc] TRODAT-1 variables | ADHD patients (n=14) |
Healthy controls (n=12) |
P-value |
|---|---|---|---|
| RC | 3.05 (2.91 , 3.46) | 2.83 (2.62 , 3.28) | 0.303 |
| LC | 3.10 ± 0.69 | 2.97 ± 0.31 | 0.835 |
| RP | 2.51 ± 0.31 | 2.34 ± 0.40 | 0.228 |
| LP | 2.38 ± 0.49 | 2.40 ± 0.37 | 0.923 |
| RM | 2.78 ± 0.41 | 2.66 ± 0.39 | 0.446 |
| LM | 2.70 ± 0.51 | 2.68 ± 0.30 | 0.928 |
Abbreviations: ADHD, attention deficit/hyperactivity disorder; DAT, dopamine
transporter availability; RC, caudate at right side; RP, putamen at right side; RM, mean
value of caudate and putamen at right side; LC, caudate at left side; LP, putamen at left
side; LM, mean value of caudate and putamen at left side.
Data were represented as mean ± SD and compared using two-sample t-test if the data
followed normal distribution or median (IQR) and were compared using a Mann-Whitney
U test if the data didn’t follow a normal distribution.
No significant differences were noted between groups.
Table 3: Comparison of DAT availability of ADHD patients and healthy controls.
Figure 1: A representative SPECT TRODAT-1 brain image.
The region of interest at the “caudate” is circled by the blue line in the upper part of the striatum. The “putamen” is circled by the red line at the lower part of the striatum. The region of interest, in red, indicates higher dopamine transporter availability.
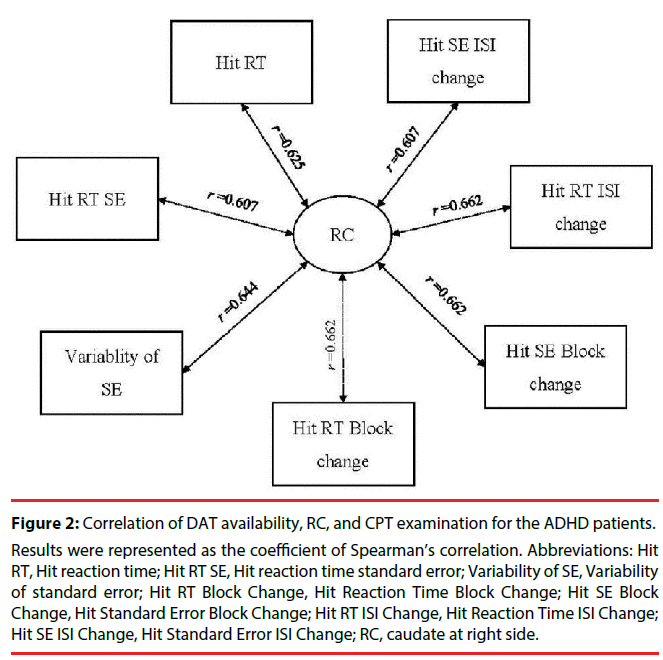
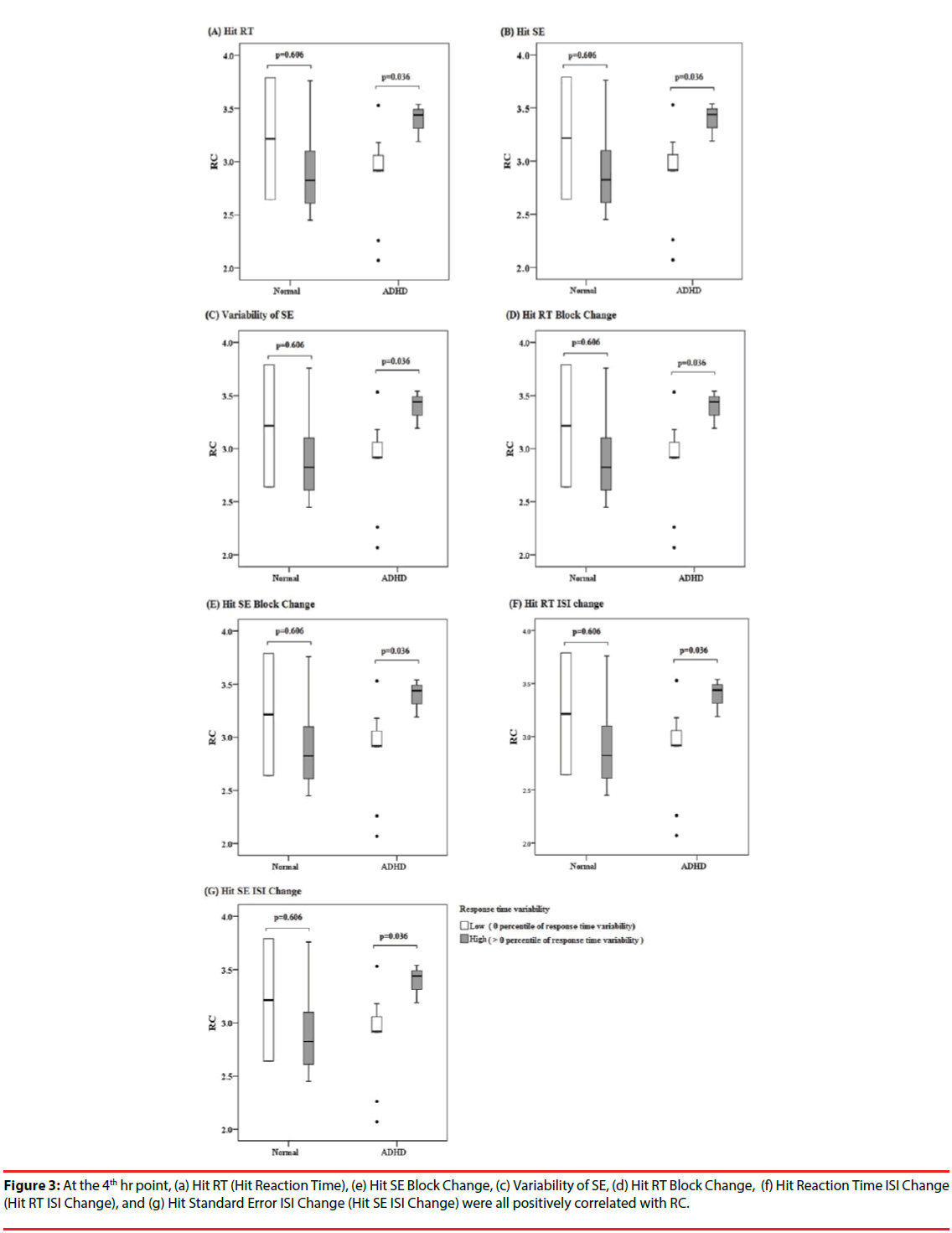
Table 4 presents the correlation of the CPT examination with DAT availability for time points for ADHD patients only. Two parameters, d-prime and beta, were not evaluated, due to the small sample size available in the ADHD group (n=4). As shown in Table 4, and Figures 2 and 3, at the 4th hr, Hit RT, Hit SE Block Change, Variability of SE, Hit RT Block Change, Hit SE Block Change, Hit Reaction Time ISI Change), and Hit Standard Error ISI Change were all positively correlated with RC.
| Omission | Commission | Hit RT | Hit RT SE |
Variability of SE |
Perservation | Hit RT Block Change |
Hit SE Block Change |
Hit RT ISI Change |
Hit SE ISI Change |
|
|---|---|---|---|---|---|---|---|---|---|---|
| RC | 0.37 | -0.074 | 0.625* | 0.607* | 0.644* | 0.180 | 0.662* | 0.662* | 0.662* | 0.607* |
| LC | 0.074 | -0.18 | -0.166 | -0.036 | 0.064 | -0.268 | -0.07 | -0.301 | -0.331 | -0.225 |
| RP | -0.012 | 0.242 | -0.069 | -0.147 | 0.239 | 0.079 | 0.528 | 0.084 | -0.022 | -0.119 |
| LP | 0.194 | -0.001 | 0.033 | 0.021 | 0.305 | -0.256 | 0.379 | -0.084 | -0.157 | -0.018 |
| RM | 0.129 | 0.093 | 0.028 | 0 | 0.326 | 0.078 | 0.45 | 0.082 | -0.004 | -0.041 |
| LM | 0.151 | -0.12 | 0.013 | -0.015 | 0.202 | -0.317 | 0.155 | -0.246 | -0.305 | -0.16 |
Abbreviations: Hit RT, Hit reaction time; Hit RT SE, Hit reaction time standard error; Variability of SE, Variability of standard error ; Hit RT Block Change, Hit Reaction Time Block Change; Hit SE Block Change, Hit Standard Error Block Change; Hit RT ISI Change, Hit Reaction Time ISI Change; Hit SE ISI Change, Hit Standard Error ISI Change; RC, caudate at right side; RP, putamen at right side; RM, mean value of caudate and putamen at right side; LC, caudate at left side; LP, putamen at left side; LM, mean value of caudate and putamen at left side.
Table 4: Correlation of DAT availability and CPT examination for ADHD patients.
Figure 2: Correlation of DAT availability, RC, and CPT examination for the ADHD patients.
Results were represented as the coefficient of Spearman’s correlation. Abbreviations: Hit RT, Hit reaction time; Hit RT SE, Hit reaction time standard error; Variability of SE, Variability of standard error; Hit RT Block Change, Hit Reaction Time Block Change; Hit SE Block Change, Hit Standard Error Block Change; Hit RT ISI Change, Hit Reaction Time ISI Change; Hit SE ISI Change, Hit Standard Error ISI Change; RC, caudate at right side.
Discussion
▪ RT variability of adults with ADHD
This is the first study to demonstrate the relationship between brain dopamine transporter availability and RT variability in young adults with ADHD. One novel finding was the association of DAT availability and RT variability in the right caudate. Thus, results from a brain examination might be used to monitor treatment response among patients with ADHD. Future studies should investigate whether enhanced dopamine transmission might be helpful to recover the deficits in executive functions, such as RT variability.
Recent research has explored the possible role of structural factors in ADHD. In a recent study, reaction time parameters during a flanker task [31] were correlated with mean diffusivity measurements in the basal ganglia and thalamus in children with ADHD and controls [32]. The results showed a correlation between mean diffusivity measurements in the deep gray matter of the striatum and thalamus and RT parameters during this visuospatial task in the controls, who were typically developing children, and in children with ADHD. Thus, microstructural changes in deep gray matter could alter the afferent/efferent signals transmitted to and from the cortex.
Alterations in the general mechanisms underlying normal brain development among patients with ADHD have also been suspect in RT variability. The caudate and putamen have important roles in several basal-ganglia-thalamocortical circuits that affect motor control and learning, as well as in selecting and enabling cognitive, executive, and emotional reactions. The results of a study of 307 persons with ADHD, 169 of their unaffected siblings and 196 typically developing controls (mean age: 17.21 yr; age range: 8-30 yr) indicated that global differences in gray matter volume may be due to changes in the general mechanisms underlying normal brain development in persons with ADHD [33]. Participants with ADHD had a 2.5% smaller total brain volume than did controls. Alterations in total gray matter and caudate and putamen volumes in unaffected siblings suggest that these volumes are linked to a familial risk for ADHD.
Karalunas, et al. [34] closely examined RT variability among participants with ADHD and autism spectrum disorders (ADS). They first performed a meta-analysis of RT variability in ADS with and without comorbid ADHD. RT variability showed some specificity to ADHD, including to a subset of children with ASD who shared a comorbid ADHD diagnosis. A second meta-analysis compared ADHD and non- ADHD children on diffusion model parameters. At the behavioral level, the authors suggest that RT variability is likely a measure of mechanisms already being investigated in children with ADHD, for example, cognitive control, arousal, and motor output problems, rather than a distinct or novel mechanism. The authors also concluded that RT variability is increased in ASD only when children with comorbid ADHD are included in the test sample.
In our study, RT variability was consistently and significantly reflected in the ADHD group’s performance on the CPT over time. Intraindividual variability, including hit reaction time, varied during the entire test. The response time was sometimes quicker and sometimes slower (shown by the slope of change in reaction time over the six time blocks, Hit Reaction Time Block Change) (Figure 2). There was a lack of consistency in performance as the test progressed (revealed in the slope of the change in reaction time standard error over the six time blocks, and Hit reaction time block standard error), and there was a loss of vigilance during the test (Hit Standard Error Block Change).
Our results also indicated that the RT variability revealed by the sub-scores over time might not be detected by commission errors on the CPT [35]. Previous studies have shown that factors other than temporal processing, inhibitory control, and delay-related deficits differentiate individuals with ADHD from controls [10]. Consistent with the results of a previous study of the moment-to-moment variability of performance [36], this suggested that the timing deficit, but not incorrect immediate responses, could differentiate those with ADHD from controls. Also consistent with the results of a previous study of children with ADHD [37], our results indicated that the adults with ADHD showed more cognitive inflexibility, represented by more perseveration, than did controls. This suggested that the deficit in executive function might occur across the life span.
▪ Response time variability and dopamine function
Brain dopamine dysfunction in ADHD could explain why stimulant medications, which increase dopamine signaling, are therapeutically beneficial. Upregulation of dopamine transporter availability during long-term treatment with methylphenidate may decrease treatment efficacy and exacerbate symptoms while patients are not under the effects of the medication. Discrepancies in the literature regarding dopamine transporter availability in ADHD participants (some studies report increases, others report no changes, and others report decreases) suggest that DAT abnormality is involved in the pathobiology that might be heterogeneous in ADHD [38].
Our results suggested that DAT availability was associated with RT variability in patients with ADHD. Previous studies have indicated that age [39] or frontal lobe injury [40], or energy deficiency [7], or impaired sensorimotor timing [41] might be associated with RT variability. However, our results showed that higher DAT availability was significantly correlated with the slowing of RT and erratic responses as the time between targets increased, and with impulsivity, random responses, and inattentiveness in the participants with ADHD. A previous study had found that abnormal dopamine transmission was associated with risk-taking, poor response inhibition, aversion to delay, and difficulty in motivation [42]. Patients who have fluctuating responses might have insufficient recruiting of brain regions associated with sensorimotor timing [6].
To the best of our knowledge, our preliminary findings are the first demonstration of the association of striatal dopamine transporter availability with RT variability in adults with ADHD. It is worth noting that a previous study had shown that striatal dopamine transporter availability was associated with executive functions in healthy young adults [43]. In addition, RT variability has been proposed as a candidate endophenotype of ADHD [44], and psychostimulants enhance RT variability in ADHD [12].Therefore, our results suggested that striatal dopamine transporter availability might be a possible biological marker to monitor RT variability in the course of treatment of adults with ADHD.
In line with the results of previous studies using 123I-β-CIT in 9 adults with ADHD [45] and 11C-PE2I in 12 adolescents [35], but in contrast to findings of other studies [35,45], the results of our study showed that compared to normal controls, treatment-naïve adults with ADHD had no difference in DAT availability in the caudate and putamen. This inconsistent finding might be due to the heterogeneity of the clinical sample. Most of the participants in our study were college graduates who were nonsmokers. None had comorbid anxiety, depression/ bipolar disorder, or substance use, oppositionaldefiant disorder, conduct disorder, or antisocial personality disorder. Our sample might therefore represent a distinct subgroup with less severe ADHD. In addition, our study was designed to control for the effects of age and smoking on DAT availability by recruiting patients under age 35. Hesse. et al. [21] had reported that the effect of age on DAT availability might not start until 35 to 40 years of age. Since participants in our study did not smoke, this might have affected DAT availability.
We also found that DAT availability was not associated with the severity of ADHD symptoms, either in inattentive or hyperactivity/impulsivity sub-scores on the CAARS. Previous studies have shown that both inattention and hyperactivity/ impulsivity were associated with altered striatal dopamine concentration [45] and DAT availability [45,46]. The lack of a significant association between clinical rating scales and DAT availability suggests that mechanisms other than DAT may be involved in the pathobiology of ADHD.
Some of our results were contradictory. Lower DAT availability was correlated with the clinical severity of ADHD, as shown by the score on the ADHD clinical index. A possible explanation is that the ADHD clinical index is a combination of all the measurements on the CPT and lacks specificity for neurocognition. In addition, lower DAT availability was correlated with the level of impulsivity, random responding, and inattentiveness represented by perseveration during the CPT in controls. A future study could have a larger sample of normal participants in order to explore the relationship between DAT availability and scores on the CPT.
Limitations
One limitation of this study was that it allowed only for subdivision of the striatum into caudate and putamen regions because of the low spatial resolution of SPECT. It was therefore difficult to detect subtle changes in other regions, including the extrastriatal areas, which might be related to the pathobiology of ADHD. We did not study all subtypes of ADHD, in order to correct the confounders in terms of sampling heterogeneity. In Kofler, et al.’s meta-analytic review, RT variability was a stable feature of ADHD and other clinical disorders [5]. Therefore, the results of our study cannot be generalized to adults with different kinds of ADHD including the comorbid with other psychiatric disorders or those exposed to psychotropic medication. Other limitations were that correlation analyses were only given for the ADHD patients, the focus of our study, and no power calculation was included.
Conclusions
RT variability is associated with abnormal brain dopamine transmission. This suggests that greater RT variability might lead adults with ADHD to have poorer and inconsistent performance. A future study to recruit more subjects to validate these preliminary results is warranted.
Funding and disclosure
None.
Acknowledgements
The study was support by TSGH-C106-103.
Conflict of interest
None to declare.
References
- Rapport MD, Kofler MJ, Alderson RM, et al. Variability of attention processes in ADHD: observations from the classroom. J. Atten. Disord 12(6), 563-573 (2009).
- Klein C, Wendling K, Huettner P, et al. Intra-subject variability in attention-deficit hyperactivity disorder. Biol. Psychiatry 60(10), 1088-1097 (2006).
- Antonini TN, Narad ME, Langberg JM, et al. Behavioral correlates of reaction time variability in children with and without ADHD. Neuropsychology 27(2), 201-209 (2013).
- Alderson RM, Rapport MD, Kofler MJ. Attention-deficit/hyperactivity disorder and behavioral inhibition: a meta-analytic review of the stop-signal paradigm. J. Abnorm. Child. Psychol 35(5), 745-758 (2007).
- Kofler MJ, Rapport MD, Sarver DE, et al. Reaction time variability in ADHD: a meta-analytic review of 319 studies. Clin. Psychol. Rev 33(6), 795-811 (2013).
- Valera EM, Spencer RM, Zeffiro TA, et al. Neural substrates of impaired sensorimotor timing in adult attention-deficit/hyperactivity disorder. Biol. Psychiatry 68(4), 359-367 (2010).
- Russell VA, Oades RD, Tannock R, et al. Response variability in Attention-Deficit/Hyperactivity Disorder: a neuronal and glial energetics hypothesis. Behav. Brain. Funct 2(1), 30 (2006)
- Sonuga-Barke EJ, Halperin JM. Developmental phenotypes and causal pathways in attention deficit/hyperactivity disorder: potential targets for early intervention? J. Child. Psychol. Psychiatry 51(4), 368-389 (2010).
- Castellanos FX, Sonuga-Barke EJ, Scheres A, et al. Varieties of attention-deficit/hyperactivity disorder-related intra-individual variability. Biol. Psychiatry 57(11), 1416-1423 (2005).
- Sonuga-Barke E, Bitsakou P, Thompson M. Beyond the dual pathway model: evidence for the dissociation of timing, inhibitory, and delay-related impairments in attention-deficit/hyperactivity disorder. J. Am. Acad. Child. Adolesc. Psychiatry 49(4), 345-355 (2010).
- Solanto MV, Gilbert SN, Raj A, et al. Neurocognitive functioning in AD/HD, predominantly inattentive and combined subtypes. J. Abnorm. Child. Psychol 35(5), 729-744 (2007).
- pstein JN, Brinkman WB, Froehlich T, et al. Effects of stimulant medication, incentives, and event rate on reaction time variability in children with ADHD. Neuropsychopharmacology 36(5), 1060-1072 (2011).
- Sjowall D, Roth L, Lindqvist S, et al. Multiple deficits in ADHD: executive dysfunction, delay aversion, reaction time variability, and emotional deficits. J. Child. Psychol. Psychiatry 54(6), 619-627 (2013).
- Yeh CB, Chou YH, Cheng CY, et al. Reproducibility of brain dopamine transporter binding with Tc-99m TRODAT-1 SPECT in healthy young men. Psychiatry. Res 201(3), 222-225 (2012).
- Spencer TJ, Biederman J, Madras BK, et al. Further evidence of dopamine transporter dysregulation in ADHD: a controlled PET imaging study using altropane. Biol. Psychiatry 62(9), 1059-1061 (2007).
- Dresel S, Krause J, Krause KH, et al. Attention deficit hyperactivity disorder: binding of [99mTc]TRODAT-1 to the dopamine transporter before and after methylphenidate treatment. Eur. J. Nucl. Med 27(10), 1518-1524 (2000).
- Yeh CB, Lee CS, Ma KH, et al. Phasic dysfunction of dopamine transmission in Tourette's syndrome evaluated with 99mTc TRODAT-1 imaging. Psychiatry. Res 156(1), 75-82 (2007).
- Costa A, Riedel M, Pogarell O, et al. Methylphenidate effects on neural activity during response inhibition in healthy humans. Cereb. Cortex 23(5), 1179-1189 (2013).
- Costa A, la Fougère C, Pogarell O, et al. Impulsivity is related to striatal dopamine transporter availability in healthy males. Psychiatry. Res 211(3), 251-256 (2013).
- Wiener M, Turkeltaub P, Coslett HB. The image of time: a voxel-wise meta-analysis. Neuroimage 49(2), 1728-1740 (2010).
- Hesse S, Ballaschke O, Barthel H, et al. Dopamine transporter imaging in adult patients with attention-deficit/hyperactivity disorder. Psychiatry. Res 171(2), 120-128 (2009).
- Nandam LS, Hester R, Wagner J, et al. Methylphenidate but not atomoxetine or citalopram modulates inhibitory control and response time variability. Biol. Psychiatry 69(9), 902-904 (2011).
- Yeh CB, Gau SS, Kessler RC, et al. Psychometric properties of the Chinese version of the adult ADHD Self-report Scale. Int. J. Methods. Psychiatr. Res 17(1), 45-54 (2008).
- Conners CK, Conners’ Continuous Performance Test II (CPT II) Ver. 5. NY: Multi-Health Systems, Inc. (2002).
- Gau SS, Chong MY, Chen TH, et al. A 3-year panel study of mental disorders among adolescents in Taiwan. Am. J. Psychiatry 162(7), 1344-1350 (2005).
- Conners CK ED, Sparrow E. Conners' adult ADHD rating scales (CAARS) technical manual. NY: Multi-Health Systems, Inc. (1999).
- Kung MP, Stevenson DA, Plossl K, et al. [99mTc]TRODAT-1: a novel technetium-99m complex as a dopamine transporter imaging agent. Eur. J. Nucl. Med 24(4), 372-380 (1997).
- Verdejo-Garcia A, Benbrook A, Funderburk F, et al. The differential relationship between cocaine use and marijuana use on decision-making performance over repeat testing with the Iowa Gambling Task. Drug. Alcohol. Depend 90(1), 2-11 (2007).
- Kung HF, Kim HJ, Kung MP, et al. Imaging of dopamine transporters in humans with technetium-99m TRODAT-1. Eur. J. Nucl. Med 23(11), 1527-1530 (1996).
- Lavalaye J, Booij J, Reneman L, et al. Effect of age and gender on dopamine transporter imaging with [123I] FP-CIT SPET in healthy volunteers. Eur. J. Nucl. Med27(7), 867-8699 (2000).
- Eriksen BA, Eriksen CW. Effects of noise letters upon the identification of a target letter in a nonsearch task. Percept. Psychotrop 16(1), 1434-1149 (1974).
- Fall S, Querne L, Le Moing AG, et al. Individual differences in subcortical microstructure organization reflect reaction time performances during a flanker task: a diffusion tensor imaging study in children with and without ADHD. Psychiatry. Res 233(1), 50-56 (2015).
- Greven CU, Bralten J, Mennes M, et al. Developmentally stable whole-brain volume reductions and developmentally sensitive caudate and putamen volume alterations in those with attention-deficit/hyperactivity disorder and their unaffected siblings. JAMA. Psychiatry 72(5), 490-499 (2015).
- Karalunas SL, Geurts HM, Konrad K, et al. Annual research review: Reaction time variability in ADHD and autism spectrum disorders: measurement and mechanisms of a proposed trans-diagnostic phenotype. J. Child. Psychol. Psychiatry 55(6), 685-710 (2014).
- Jucaite A, Fernell E, Halldin C, et al. Reduced midbrain dopamine transporter binding in male adolescents with attention-deficit/hyperactivity disorder: association between striatal dopamine markers and motor hyperactivity. Biol. Psychiatry 57(3), 229-238 (2005).
- Aase H, Sagvolden T. Moment-to-moment dynamics of ADHD behaviour. Behav. Brain. Funct 1(1), 12 (2005).
- Rubia K, Cubillo A, Smith AB, et al. Disorder-specific dysfunction in right inferior prefrontal cortex during two inhibition tasks in boys with attention-deficit hyperactivity disorder compared to boys with obsessive-compulsive disorder. Hum. Brain. Mapp 31(2), 287-299 (2010).
- Wang GJ, Volkow ND, Wigal T, et al. Long-term stimulant treatment affects brain dopamine transporter level in patients with attention deficit hyperactive disorder. PLoS. One 8(1), e63023 (2013).
- Williams BR, Hultsch DF, Strauss EH, et al. Inconsistency in reaction time across the life span. Neuropsychology 19(1), 88-96 (2005).
- Stuss DT, Murphy KJ, Binns MA, et al. Staying on the job: the frontal lobes control individual performance variability. Brain 126(11), 2363-2380 (2003).
- McGough JJ, Barkley RA. Diagnostic controversies in adult attention deficit hyperactivity disorder. Am. J. Psychiatry 161(11), 1948-1956 (2004).
- Pattij T, Vanderschuren LJ. The neuropharmacology of impulsive behaviour. Trends. Pharmacol. Sci 29(4), 192-199 (2008).
- Burke SM, van de Giessen E, de Win M, et al. Serotonin and dopamine transporters in relation to neuropsychological functioning, personality traits and mood in young adult healthy subjects. Psychol. Med 41(2), 419-429 (2011).
- Johnson KA, Kelly SP, Bellgrove MA, et al. Response variability in attention deficit hyperactivity disorder: evidence for neuropsychological heterogeneity. Neuropsychologia 45(4), 630-638 (2007).
- Volkow ND, Wang GJ, Newcorn J, et al. Brain dopamine transporter levels in treatment and drug naive adults with ADHD. Neuroimage 34(3), 1182-1190 (2007).
- Gainetdinov RR, Caron MG. Monoamine transporters: from genes to behavior. Annu. Rev. Pharmacol. Toxicol 43(1), 261-284 (2003).